Published on
Urgent Message: Deep tissue infections are possible considerations for diabetic patients presenting to urgent care with muscle tenderness who recently have had joint or bursal aspirations or injections.
Citation: Mati M. Quadriceps Pyomyositis After Aspiration of Patellar Bursitis: A Case Report. J Urgent Care Med. 2024;18(4):25-27.
Key Words: quadriceps myositis, diabetes, patellar bursitis, infectious disease
Abstract
Introduction
Infectious myositis and pyomyositis are uncommon and therefore easily overlooked with serious potential complications of bursal aspirations.
Clinical Findings
A 56-year-old male with medical history of moderately controlled diabetes, dyslipidemia, and hypertension presented to urgent care (UC) with several weeks of worsening thigh pain. Two months prior to this presentation, he had a patellar bursal aspiration, was diagnosed with septic bursitis, and was treated with oral antibiotics. He had exquisite tenderness to palpation. Point of care ultrasound (POCUS) did not show signs of deep vein thrombosis (DVT), fluid collections, or evidence of cellulitis. Labs were remarkable for elevated white blood cell count (WBC) and C-reactive protein (CRP). CT scan showed concern for myositis.
Diagnosis
The patient was sent to the emergency department for magnetic resonance imaging (MRI) and orthopedic consultation. He was admitted to the hospital, underwent surgical drainage and debridement, completed 4 weeks of IV antibiotics, and ultimately fully recovered.
Conclusion
Clinicians are encouraged to consider deep tissue infections in diabetic patients who recently have had joint or bursal aspirations or injection.
Introduction
This case illustrates an uncommon potential complication of joint or bursal aspiration: infectious myositis. It follows the trajectory of the patient, outlines data points that led to the diagnosis, and reminds the reader about the pitfalls of some cognitive biases.
Patient Information
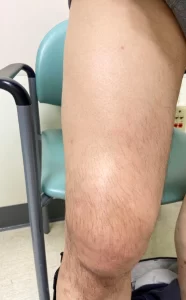
A 56-year-old male with a past medical history of diabetes mellitus, hypertension, and hyperlipidemia presented to the urgent care (UC) with a contusion to his right knee with pain, swelling, and decreased range of motion. He underwent a prepatellar bursal aspiration, was diagnoses with septic bursitis, and was started on trimethoprim-sulfamethoxazole. Several days later, the aspirate cultures grew back methicillin-susceptible Staphylococcus aureus (MSSA). On follow up 3 weeks later, he confirmed completion of the antibiotic course and resolution of symptoms.
He returned to the UC 7 weeks later with 7 days of worsening swelling, pain, and tenderness to his right anterior thigh. He went to his primary care provider who diagnosed a muscle strain and prescribed naproxen.
He was unable to bear weight on his right leg and had swelling to the anterior thigh. He denied pain to knee, groin, or lower leg. He denied fevers, chills, nausea, or vomiting. He did not experience recent long distance travel, recent surgery, or a personal or family history of blood clots. Likewise, he experienced no new traumatic events.
Clinical Findings
On exam, the patient was non-toxic appearing, afebrile, with a heart rate of 76 and blood pressure of 135/68. His right thigh appeared larger compared to his left without any erythema or skin changes. There was an area of approximately 4cm x 4cm of non-fluctuant induration on the anterior thigh that was very tender to touch. He had full range of motion. Sensation and pulses were intact. There was an enlarged, mildly tender inguinal region lymph node.
Diagnostic Assessment
Labs collected from urgent care were unremarkable other than a WBC of 10.4 x 103/uL and a CRP of 25 mg/dL. Point of care ultrasound showed no blood clots along the femoral vein into the popliteal, no knee joint effusion, however, a small suprapatellar effusion was noted. A large lymph node was noted in the inguinal area as well.
A CT scan was obtained, which showed a large region of intermediate density along the rectus femoris muscle and tendon with areas of fluid along the fascial planes throughout the thigh. The patient was sent to the emergency department for an MRI and admission. An MRI showed intramuscular abscess in the anterior compartment of the thigh with adjacent muscular edema.
The patient was diagnosed with pyomyositis, started on IV vancomycin after blood cultures were collected, and admitted to the hospital.
Therapeutic Intervention
In the operating theater, a large area of purulence was found in the anterior thigh which communicated with the prepatellar area. Ultimately, all cultures grew back MSSA. The patient was discharged with a peripherally inserted central catheter (PICC) and cefazolin for 4 weeks.
Follow-up and Outcomes
The patient completed his course of antibiotics without complications. Physical therapy helped him return to baseline functional status.
Discussion
Infectious myositis is inflammation of muscle caused by an infectious agent.1 Pyomyositis is an infection of skeletal muscle and is frequently associated with abscess formation.2 Whereas primary pyomyositis refers to cases when there is no obvious local source of infection and hematogenous spread is assumed,3 secondary pyomyositis refers to instances when there is contiguous spread or local penetrating wound.4
This case is interesting in that the presumed inciting incident—the bursal aspiration—was many weeks prior to the onset of symptoms. This delay in onset raises the potential for a primary pyomyositis. Risk factors for primary pyomyositis include conditions where the immune system is suppressed such as HIV/AIDS, diabetes mellitus (DM), organ transplant, and chronic kidney disease; other risk factors include muscle overuse, blunt trauma, malnutrition, and skin conditions (eg, eczema or varicella).2,5–8
In our case, MRI and surgical findings point to a secondary pyomyositis. The primary causative infectious agent in both primary and secondary pyomyositis is most commonly Staphylococcus aureus.9 In our case, all cultures grew back MSSA. There is often a 3-stage progression noted in pyomyositis: an insidious phase lasting days to weeks; abscess formation; and, if untreated, potential septic shock due to bacteremia.5 It is possible that our patient had an infectious myositis partially treated by his initial oral antibiotics then had a prolonged initial phase with eventual abscess formation.
The case illustrated the high index of suspicion often needed to make this diagnosis early, as exam findings can be subtle.6 Our patient had pain without evidence of cellulitis or DVT. It would have been easy to dismiss his complaints, as occurred with the primary care clinician, knowing muscle strain may trigger pyomyositis.10
However, given his comorbid DM, relatively recent bursal aspiration, enlarged lymph node, exquisite tenderness to palpation, and negative POCUS for DVT, cellulitis, or fluid collection, further workup was pursued. POCUS identification of fluid collecting within fascial planes is indicative of pyomyositis and a signal to further investigate with MRI.11 In our case, POCUS was used to make other diagnoses on the differential less likely, specifically DVT, cellulitis, and subcutaneous abscess.
Obtaining labs and noting an elevated CRP further increased our suspicion for an underlying inflammatory process such as infection. Ultimately, recovery of purulent fluid in the operating room provided the definitive diagnosis, but CT scan with IV contrast was easily available in our urgent care and provided enough evidence to admit to the hospital and start IV antibiotics.
Patient Perspective
The patient was unhappy spending so much time in the hospital and rehabilitating from the surgical incision and debridement. But he was very appreciative of the care he received in the urgent care. Specifically, he was grateful for being listened to and having his symptoms taken seriously.
Ethics Statement
Consent to publish the case was verbally obtained over the phone by speaking to the patient directly.
Manuscript submitted September 11, 2023; accepted November 22, 2023.
Author Affiliation: Benjamin Mati, MD, Ventura County Medical Center, Department of Emergency Medicine, Department of Critical Care Medicine. Author has no relevant financial relationships with any ineligible companies.
References
- Radcliffe C, Gisriel S, Niu YS, Peaper D, Delgado S, Grant M. Pyomyositis and Infectious Myositis: A Comprehensive, Single-Center Retrospective Study. Open Forum Infect Dis. 2021;8(4):ofab098. doi:10.1093/ofid/ofab098
- Ngor C, Hall L, Dean JA, Gilks CF. Factors associated with pyomyositis: A systematic review and meta‐analysis. Trop Med Int Health. 2021;26(10):1210-1219. doi:10.1111/tmi.13669
- Marath H, Yates M, Lee M, Dhatariya K. Pyomyositis. J Diabetes Complications. 2011;25(5):346-348. doi:10.1016/j.jdiacomp.2010.09.002
- Stevens DL, Bisno AL, Chambers HF, et al. Executive Summary: Practice Guidelines for the Diagnosis and Management of Skin and Soft Tissue Infections: 2014 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59(2):147-159. doi:10.1093/cid/ciu444
- Agarwal V, Chauhan S, Gupta RK. Pyomyositis. Neuroimaging Clin N Am. 2011;21(4):975-983. doi:10.1016/j.nic.2011.07.011
- Habeych ME, Trinh T, Crum-Cianflone NF. Purulent infectious myositis (formerly tropical pyomyositis). J Neurol Sci. 2020;413:116767. doi:10.1016/j.jns.2020.116767
- Maravelas R, Melgar TA, Vos D, Lima N, Sadarangani S. Pyomyositis in the United States 2002–2014. J Infect. 2020;80(5):497-503. doi:10.1016/j.jinf.2020.02.005
- Small LN, Ross JJ. Tropical and Temperate Pyomyositis. Infect Dis Clin North Am. 2005;19(4):981-989. doi:10.1016/j.idc.2005.08.003
- Bickels J, Ben-Sira L, Kessler A, Wientroub S. PRIMARY PYOMYOSITIS: J Bone Jt Surg-Am Vol. 2002;84(12):2277-2286. doi:10.2106/00004623-200212000-00024
- Lovejoy JF, Alexander K, Dinan D, Drehner D, Khan-Assad N, Lacerda IRA. Team Approach: Pyomyositis. JBJS Rev. 2017;5(6):e4-e4. doi:10.2106/JBJS.RVW.16.00048
- Levitt DL, Byer R, Miller AF. Point-of-Care Ultrasound to Diagnose Pyomyositis in a Child: Pediatr Emerg Care. 2019;35(1):69-71. doi:10.1097/PEC.0000000000001711
Download the article PDF: Quadriceps Pyomyositis After Aspiration of Patellar Bursitis: A Case Report
Read More
