Published on
Urgent message: Proptosis may be caused by a variety of vascular, infectious, endocrine, and neoplastic diseases that may threaten vision or cause serious systemic complications or death. Providers should be familiar with causes and work-up, including which findings suggest a need for immediate imaging and consultation.
ARMIN GOLLOGLY, MD, BRIAN ZIPSER, MD, WENDY HUANG, MD, FAAO, FAAP,
CHRISTOPHER M. FECAROTTA, MD, FAAO, FAAP, FCPP, AND MARK RICHMAN, MD, MPH
Introduction
This report describes a case of unilateral proptosis due to an intraorbital arteriovenous malformation. Proptosis can be caused by a variety of vascular, infectious, endocrine, and neoplastic diseases that may threaten vision or cause serious systemic complications or death. Accurate diagnosis and timely and appropriate referral from the urgent care setting can prevent these complications. Thus, it is important for urgent care providers to understand the disease processes that can present with proptosis and include them in the differential diagnosis when evaluating eye problems.
Case Presentation
A 32-year-old man presented to an urgent care clinic with a 5-year history of right-eye pain and gradual swelling. There had been no inciting trauma, fever, or hyperthyroid symptoms. He had no blurry or double vision. There was no family history of ocular problems.
Physical Examination
On initial presentation, the patient’s vital signs were as follows:
- Temperature: 5°C
- Blood pressure: 134/90 mm Hg
- Pulse: 83 beats/min
- Respiratory rate: 19 breaths/min
- Oxygen saturation: 99% on room air
On physical examination, the patient appeared to be well. He demonstrated right-eye injection, proptosis, and upper-lid swelling. His pupils were equally round and reactive to light; extraocular movements were intact. His dentition was unremarkable, and his sinuses were not tender. His visual acuity was 20/20 bilaterally with- out correction.
Imaging
The patient was referred for magnetic resonance imaging (MRI).
Diagnosis
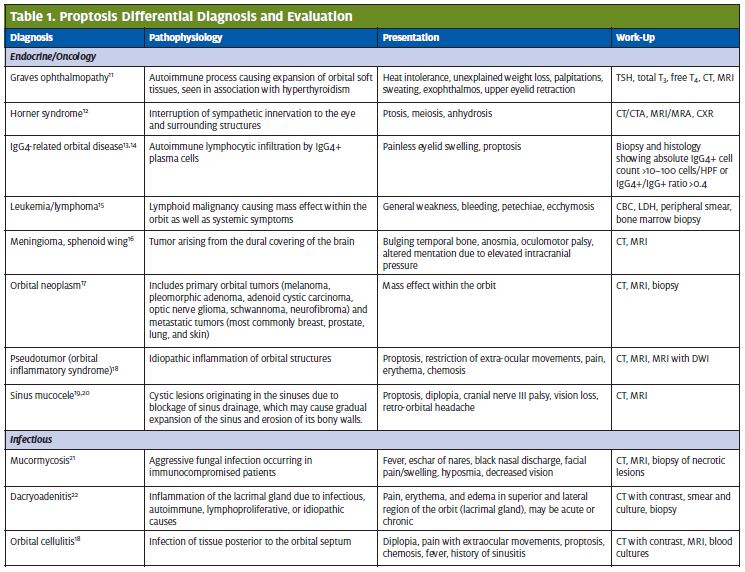
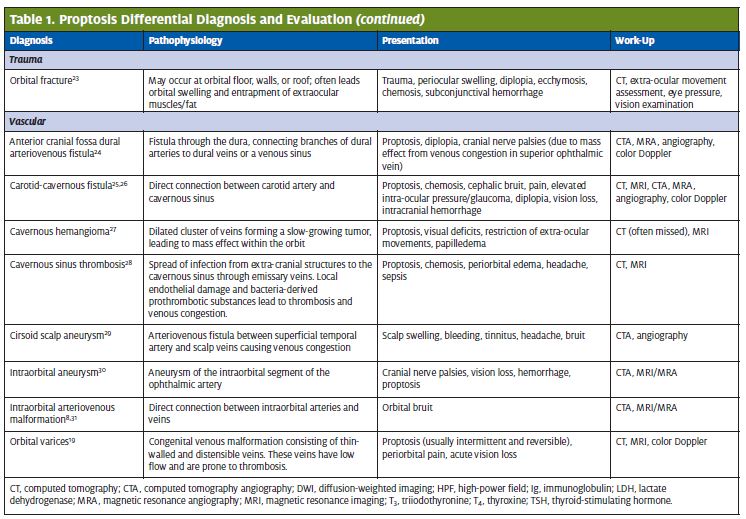
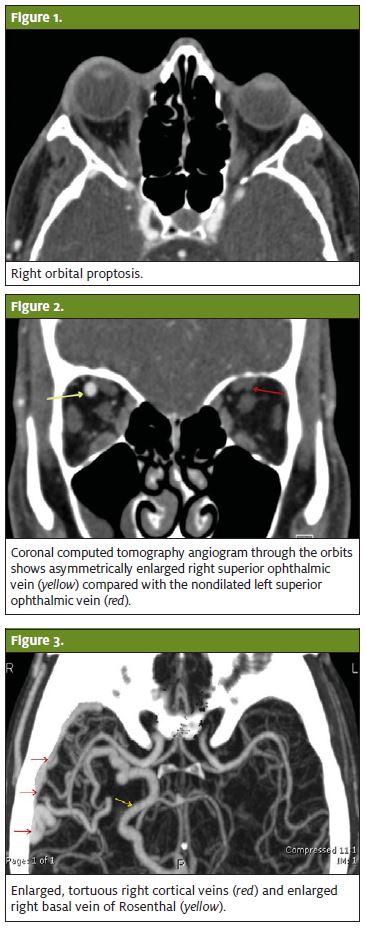
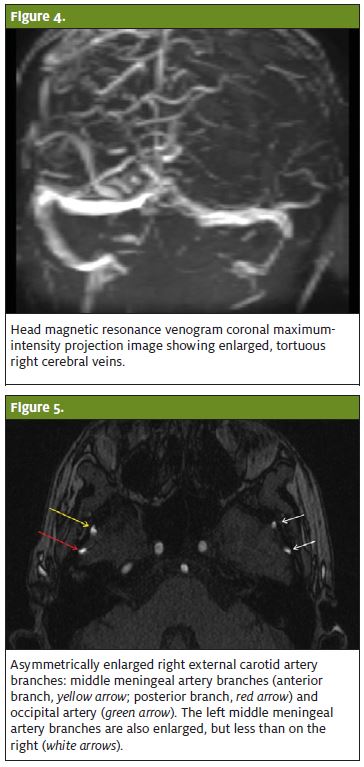
The diagnosis was arteriovenous malformation (AVM), as seen in Figures 1–5, rather than carotid-cavernous fistula.
 Discussion
Discussion
Proptosis is an uncommon presentation or examination finding, with an incidence of 16 cases/100,000 persons per year among women and 2.9 cases/100,000 persons per year among men.1 Proptosis can be subtle, and thus it may go unnoticed by the patient if it develops gradually. In the evaluation of eye presentations, especially with concerning features such as diplopia, vision loss, and restriction of extraocular movements, proptosis is an important physical examination finding that can help direct the work-up. Proptosis is easily missed, but looking for it can lead to timely imaging and treatment before the underlying disease irreversibly damages the patient’s eyesight.
Anatomy
Blood flow to the eye is supplied by the internal carotid artery, which gives rise to the ophthalmic artery, which then branches into the central retinal artery and other smaller vessels. Venous drainage occurs through the central retinal vein and vortex veins; these drain into the cavernous sinus, the pterygoid venous plexus, and the facial vein.2 AVMs occur when the embryonic vasculature does not differ- entiate in one or more locations, leading to a mass of tangled arteries and veins. The resulting high-pressure shunt between the arterial and venous systems causes congestion in the veins draining the AVM, leading to mass effect, tissue edema, and aneurysm formation.3 In both fistulas and malformations, blood bypasses the capillaries and tissues: Arteriovenous fistulas involve a single connection between one artery and one vein, whereas AVMs involve multiple connections.4
Diagnosis and Disposition
When evaluating proptosis, clinicians must consider a broad differential diagnosis, ranging from benign to sight- and life-threatening conditions (Table 1). Patients with unilateral proptosis should have same-day or urgent orbital imaging with intravenous contrast. If imaging suggests a vascular lesion, follow-up imaging should include contrast evaluation of cranial vasculature. Initial treatment of AVMs can be done medically, including symptomatic management of headache and control of hypertension and diabetes mellitus.5 Once identified, AVMs should be assessed in a neurosurgery department or a neuro-interventional radiology department for definitive surgical treatment.6
Treatment
Surgical options for the treatment of intraorbital AVM include micro- surgical resection to remove the nidus of the AVM, endovascular embolization with thrombosing agents to reduce the size of the shunt, and stereotactic radiosurgery, which also reduces the shunt via radiation-induced thrombosis. Endovascular embolization is the preferred, least-invasive procedure, with a high cure rate and mini- mal risks. These three modalities may also be used in combination to maximize effect or reduce risk of hemorrhage; usually preopera- tive embolization is followed by surgical excision.7 Whichever treat- ment strategy is chosen, long-term follow-up is recommended to assess for regrowth of the lesion.
Complications
Major complications of intraorbital AVMs include intraorbital hemorrhage, headaches, eye pain, glaucoma, and visual disturbances such as decreased visual acuity (if cranial nerve II is affected) and diplopia (if cranial nerves III, IV, and/or VI are affected).6 The mortality rate for AVM-related hemorrhage is 6% to 14%, and the annual risk of bleeding for patients with an AVM is 1% to 2%.8
Sudden onset of severe pain, nausea and vomiting, worsening proptosis, decreased visual acuity, and restriction of extraocular movements in the setting of a known or suspected AVM may rep- resent orbital hemorrhage and retrobulbar hematoma. Bleeding within the confined space of the orbit leads to an orbital compartment syndrome and retinal ischemia, causing irreversible vision loss in as little as 60 to 100 minutes.9 There are few reported cases detailing the treatment of acute spontaneous hemorrhage of AVMs; lateral canthotomy and cantholysis have been used in postoperative AVM bleeds.10 Decompression by lateral canthotomy should be considered when there is evidence of intraocular compartment syndrome, as evidenced by a tonometric measurement of intraoc- ular pressure >40 mm Hg.9
 Take-Home Points
Take-Home Points
The patient presented in this case had slowly progressive unilateral proptosis without fever or clinical evidence of cancer, making infection or malignancy less likely. Nor- mal findings on thyroid studies excluded Graves oph- thalmopathy. Structural or vascular etiologies remained prominent on the differential diagnosis; a vascular etiol- ogy was confirmed by contrast-enhanced MRI. The patient was referred to a neuro-interventional radiology department for evaluation.
For ill-appearing patients with proptosis, indications for transfer to an emergency department include intraorbital hemorrhage, infection, and trauma. Rapid progression of symptoms, severe pain, and decreased vision may indicate intraorbital hemorrhage and retrobulbar hematoma, which should be managed by lateral canthotomy and by transfer to facilities with ophthalmology services. Patients for whom there is high suspicion for infection, including rapid progression, pain, fever, or risk factors for mucormycosis, may be transferred with intravenous antibiotics or antifungals.
In contrast, patients with slowly progressive proptosis not suggestive of infection or trauma can safely be dis- charged for semi-urgent imaging for vascular or structural abnormalities as long as close follow-up can be ensured. Ultimately, these clinically well patients may need referral to ophthalmology, neurosurgery, or neurointerventional radiology departments, depending on imaging results.
- Bartley GB, Fatourechi V, Kadrmas EF, et The incidence of Graves’ ophthalmopathy in Olmstead County, Minnesota. Am J Ophthalmol. 1995;120:511–517.
- Rootman J, Heran MKS, Graeb Vascular malformations of the orbit: classification and the role of imaging in diagnosis and treatment strategies. Ophthal Plast Reconstr Surg. 2014;30:91–104.
- Moin M, Kersten RC, Bernardini F, et Spontaneous hemorrhage in an intraorbital arteriovenous malformation. Ophthalmology. 2000;107:2215–2219.
- Rowh AD, Ufberg JW, Chan TC, et Lateral canthotomy and cantholysis: emergency management of orbital compartment syndrome. J Emerg Med. 2015;48:325–330.
- Levin MH, Moss HE, Pineles SL, et Orbital congestion complicating treatment of cerebral vascular anomalies. World Neurosurg. 2014;82(1/2):239.e13–239.e17.
- Bahn Graves’ ophthalmopathy. N Engl J Med. 2010;362:726–738.
- Walton KA, Buono Horner syndrome. Curr Opin Ophthalmol. 2003;14:357–363.
- Andrew N, Kearney D, Selva IgG4-related orbital disease: a meta-analysis and review.
- Biousse V, Mendicino ME, Simon DJ, et The ophthalmology of intracranial vascular abnormalities. Am J Ophthalmol. 1998;125:527–544.
- Rootman J, Heran MKS, Graeb Vascular malformations of the orbit: classification and the role of imaging in diagnosis and treatment strategies. Ophthal Plast Reconstr Surg. 2014;30:91–104.
- Mohr JP, Parides MK, Stapf C, et al; international ARUBA Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicenter, non-blinded, randomised trial. Lancet. 2014;383: 614–621.
- Warrier S, Prabhakaran VC, Valenzuela A, et Orbital arteriovenous malformations.Hayreh Orbital vascular anatomy. Eye. 2006;20:1130–1144. Arch Ophthalmol. 2008;126:1669–1675. Acta Ophthalmol. 2013;91:694–700.
- Islam AD, Selmi C, Ditta-Mitra A, et The changing faces of IgG4-related disease: Clinical manifestations and pathogenesis. Autoimmun Rev. 2015;14:914–22.
- Bernardini FP, Bazzan M. Lymphoproliferative disease of the orbit. Curr Opin Ophthalmol. 2007;18:398–401.
- Whittle IR, Smith C, Navoo P, et Meningiomas. Lancet. 2004;363:1535–1543.
- Tailor TD, Gupta D, Dalley RW, et Orbital neoplasms in adults: clinical, radiologic, and pathologic review. Radiographics. 2013;33:1739–1758.
- Pakdaman MN, Sepahdari, AR, Elkhamary Orbital inflammatory disease: pictorial review and differential diagnosis. World J Radiol. 2014;6:106–115.
- Pahwa S, Sharma S, Das CJ, et Intraorbital cystic lesions: an imaging spectrum. Curr Probl Diagn Radiol. 2015;44:437–448.
- Fernandes R, Pirgousis Longstanding giant frontal sinus mucocele resulting in extra- axial proptosis, orbital displacement, and loss of vision. J Oral Maxillofac Surg. 2010;68:3051–3052.
- Lemos J, Eggenberger Neuro-ophthalmological emergencies. Neurohospitalist. 2015;5:223–233.
- Mombaerts The many facets of dacryoadenitis. Curr Opin Ophthalmol. 2015;26:399–407.
- Boyette JR, Pemberton JD, Bonilla-Velez Management of orbital fractures: challenges and solutions. Clin Ophthalmol. 2015;9:2127–2137.
- Hacein-Bey L, Konstas AA, Pile-Spellman Natural history, current concepts, classi- fication, factors impacting endovascular therapy, and pathophysiology of cerebral and spinal dural arteriovenous fistulas. Clin Neurol Neurosurg. 2014;121:64–75.
- Korkmazer B, Kocak B, Tureci E, et Endovascular treatment of carotid cavernous sinus fistula: a systematic review. World J Radiol. 2013;5:143–155.
- Rose GE, Verity Acute presentation of vascular disease within the orbit—a descrip- tive synopsis of mechanisms. Eye. 2013;27:299–307.
- Scheuerle AF, Steiner HH, Kolling G, et Treatment and long-term outcome of patients with orbital cavernomas. Am J Ophthalmol. 2004;138:237–244.
- Desa V, Green Cavernous sinus thrombosis: current therapy. J Oral Maxillofac Surg. 2012;70:2085–2091.
- Goel V, Verma AK, Singh S, et Cirsoid aneurysm of scalp: demonstration on CT angiography (CTA). BMJ Case Rep. Published online: 2013. Accessed: 16 January 2016. doi: 10.1136/bcr-2013-202459.
- Della Pepa GM, Sabatino G, La Rocca G, et Intraorbital and intracanalicular oph- thalmic artery aneurysms. Literature review and report of a case. Br J Neurosurg. 2014;28:787–790.
- Hieu PD, Besson G, Roncin S, Nonent Successful surgical treatment of intraorbital arteriovenous malformations: case report. Neurosurgery. 1997;40:626–631.