Published on
Urgent message: Upper respiratory infections (URIs) are the most common presenting complaint in urgent care. Regardless of etiology or provider specialty, antibiotics are prescribed 60% of the time for the treatment of URIs, contributing to drug-resistant respiratory organisms. Employing a multimodal intervention, the authors we were able to appreciate a modest, statistically significant decrease in the rate of antibiotic prescribing among urgent care providers.
Introduction
Antimicrobial resistance is arguably one of the greatest risks to human health. Multidrug resistant organisms are increasingly providing clinical management challenges to providers in ambulatory and inpatient settings—often, as a result of the improper use of antibiotics in the outpatient setting.1,2 Drug-resistant organisms are hazardous to the United States public health system and have a tremendous impact on the morbidity and mortality rates for infectious diseases. Unfortunately, the development of new antibiotic agents to combat these organisms has decreased in the past few years due to the minimized return on investments by pharmaceutical companies.2,3 As a result, drug-resistant infectious diseases claim the lives of at least 23,000 people annually in the U.S.4
To address this issue in the U.S., President Obama issued Executive Order 13676 in 2014, with a national strategy to reduce the use of antibiotics in both inpatient and outpatient settings.5 Regardless of specialty, providers should practice the judicious use of antibiotics to preserve their effectiveness and prevent the emergence of a pre-antibiotic era.
Due to the overwhelming growth and utilization of urgent care centers (UCCs) in the U.S., urgent care (UC) providers have an opportunity to minimize the progression of antimicrobial resistance through the appropriate use of antibiotics when treating common infections. UCCs provide care to >71 million patients annually in the U.S., with a large percentage of visits being for upper respiratory infection (URI) complaints.6 URIs such as nasopharyngitis, rhinosinusitis, pharyngitis, and bronchitis are usually viral in nature; however, it is common for healthcare providers to prescribe antibiotics for these diagnoses. It is estimated that antibiotics are prescribed 60% of the time for URIs regardless of etiology.7 These practices can impact patient outcomes and quality of care—and contribute to antimicrobial resistance.
Similar to global trends, our hospital-owned UCCs located in two suburban communities in the Mid-Atlantic region of the U.S. experienced high rates of antibiotic prescribing for URIs. Differences in provider type (physicians; physician assistants [PAs]; nurse practitioners [NPs]), knowledge base, confidence, and practice experiences, along with the lack of company-adopted treatment guidelines are possible contributors. A quality improvement initiative was implemented to standardize provider care through the use of evidence-based practice (EBP) guidelines for URI management. In this article, we will discuss how EBP interventions were translated into practice to reduce inappropriate antibiotic prescribing for URIs in the UC setting.
The Literature Review
A systematic review of the literature was conducted to discover interventions that demonstrated statistical significance in reducing antibiotic use for URI management in UCCs. Studies targeting UCCs were scarce; most occurred in primary care settings. Researchers measured guideline adherence by the reduction of antibiotic use for viral infections, and an increased use of first-line therapies for bacterial illnesses.8-10 When used in varying combinations, provider education, consensus meetings, algorithm use, prescriber feedback/audit, clinic champions, and patient education demonstrated effectiveness. Of these, provider education, and prescriber feedback and audit were the most commonly used.
Interventions were most commonly measured by their ability to reduce the overall rate of antibiotic prescribing or the proportion of antibiotic prescriptions for URIs.7-13 A 10% relative reduction in the rate of antibiotic prescriptions for URIs among the intervention groups, compared with control sites, demonstrated statistical significance for some studies.12,13 We selected the most commonly used components from successful programs and considered their statistical significance, implementation time, feasibility, and the practice culture.
Methods
We conducted a quality improvement project for two hospital-owned UCCs with annual volumes of 26,800. This project was reviewed by the Johns Hopkins Medicine Institutional Review Board and deemed Not Human Subject Research. The intervention included: monthly 1- hour provider consensus meetings; review of clinical guidelines4,7,14 for URI diagnosis of bronchitis, nasopharyngitis, sinusitis, or URI‒not otherwise specified (URI-NOS); clinical pathway development for sinusitis; monthly prescriber feedback/audit; and patient education. Our aims were 1) to decrease the use of antibiotics for URIs at the urgent care centers by 10% and 2) to attain an 80% utilization rate of developed guidelines for URIs by the providers.
Selection and Description of Participants
The project team consisted of two NPs, two PAs, and three physicians who participated on a voluntary basis. The intervention targeted all providers employed by the UCCs (N=10) and patients diagnosed with bronchitis, nasopharyngitis, sinusitis, or URIs between October 1, 2014 and February 1, 2015. This timeframe was chosen based on prior studies and a historical analysis of the frequency of URI encounters at the centers over the past 3 years.11,15,16
Study Design
Team meetings were open to everyone and held monthly from October 1, 2014 through February 28, 2015. These meetings sought to facilitate compliance with the best practices in the management of URIs through review of evidence-based literature, compliance coaching, and problem solving. Each 1-hour meeting was agenda-driven. Agenda items included: the purpose, goals, and timeline of the project; impact of practice variances and antibiotic overuse; review of clinical guidelines for URIs; general information on provider compliance with guidelines, and a discussion on the perceived successes and challenges of the project.
Using a consensus approach, project team members had an opportunity to review EBP guidelines from the CDC, AAFP and IDSA for bronchitis, sinusitis, and URI management. Team members were asked to adopt or adapt guidelines to best meet the needs of patients and to facilitate compliance. Upon consensus, these guidelines were adopted and clinical pathways were developed for each diagnosis using the American Heart Association standards for clinical pathway development to promote rigor and trustworthiness.17 The developed pathways and adopted clinical guidelines were made available to all providers (during the provider meeting, through company email, and displayed at each provider work station) for use starting on October 1, 2014. These documents were accompanied by a policy and procedure for use, and details on the prescriber feedback and audit methods.
Provider compliance with clinical guidelines and pathways for URIs was assessed through monthly chart audits. Treatment plans of patients with sinusitis, bronchitis and URI-NOS were compared to the clinical guidelines and pathways to determine compliance. Based on this review, all clinicians were provided with monthly confidential, timely, direct, and written feedback via email by the project manager regarding the project measures during the intervention period of October 1, 2014 through February 1, 2015. The feedback tool was adapted from a study measuring similar outcomes.8 De-identified aggregate data regarding prescription rates and guideline adherence were presented during the monthly team meetings.
The final component of this intervention targeted patients. The educational campaign provided verbal, written, and video messages/materials on the rationale for, and benefits of, appropriate antibiotic use to patients and the community. The campaign began on October 1, 2014 using exit care instructions from the electronic medical record and company-approved materials from the CDC’s Get Smart Know When Antibiotics Work (Get Smart) campaign.
Upon discharge, exit care instructions were given to each patient regarding their diagnosis from the Practice Velocity electronic medical record (EMR). When appropriate, an additional instruction page on antibiotic nonuse was included. Exit care instructions complemented education from the provider, and outlined the common causes of the diagnosis, warning signs, and information on follow up. Get Smart materials were displayed at the centers and throughout the community.
Materials and mechanisms for marketing the campaign included Get Smart campaign posters and brochures, which were displayed throughout each center; participation as a CDC Get Smart partner; radio and publication interviews for the community; an article about the project targeting all providers in the hospital system; the center’s health blog; and a video regarding the smart use of antibiotics on the center’s website. Social media marketing on Facebook and Twitter shared facts on the smart use of antibiotics.
Data Collection
Baseline prescribing rates were obtained through a retrospective chart review of 273 patient encounters with URI diagnosis of nasopharyngitis, bronchitis, sinusitis, or URI-NOS during the intervention period. The sample size calculation was based on a 12% reduction in antibiotic prescriptions for URIs in a prior study.18 A sample size of 273 per group was determined using this effect size, with a type I error of .05, and 80% power.
Inclusion criteria included patients of all ages with one of the primary International Classification of Diseases 9th (ICD9) Revision codes listed in Table 1. A patient demographic report by ICD9 code was obtained using PVM (the Practice Velocity practice management system). A systematic randomization of the sample was conducted through the selection of every third chart under each ICD9 code from the report. Each selected chart was assigned a de-identified encounter number. Charts were obtained from Velocidoc, the Practice Velocity EMR, and reviewed using the chart audit tool. The tool was deemed reliable following a pilot of 20 charts. Exclusion criteria included: a concurrent diagnosis of influenza; comorbid conditions of COPD, diabetes, cancer, or immunosuppression; patients presenting for follow up within 1 week of initial encounter; oral antibiotic use within past 30 days; and patients with other diagnoses requiring oral antibiotic use, such as urinary tract infections. Demographic information including age, sex, and gender were collected in addition to the month and year of the encounter, smoking status, ICD9 code(s), provider type, and whether an antibiotic was prescribed. A password-protected database was created using Microsoft Excel to store de-identified data.

To evaluate the intervention period, we reviewed 273 patient URI encounters using the exact methods described above. This review required the collection of additional data, including the treating provider and whether the treatment plan demonstrated adherence to the adopted guidelines. Provider names were de-identified to ensure confidentiality prior to entry into the database.
Statistical Methods
The Microsoft Excel database was exported into the Statistical Package for the Social Sciences (SPSS) 22 for analysis. Data were sorted by time period (baseline and intervention), and frequencies were conducted on each variable to ensure the completeness of data prior to performing statistical tests. For the primary outcome variable, a 10% decrease in the rate of antibiotic prescribing for URIs, the chi square test of proportions ( was used to compare prescribing rates between project periods. For the secondary outcome variable, attainment of an 80% utilization rate of URI guidelines, the frequency of guideline adherence was analyzed.
Providers were given feedback on the primary and secondary outcomes based on the analysis conducted for both outcomes using . These rates were compared for each provider to the overall group for the time period reviewed.
Further analysis included an independent sample t-test to compare the mean ages of the sample between time periods, and was used to explore differences between subgroups and the outcome variables. Statistical significance was determined by p values <0.05 with a confidence interval set at 95%.
Results
During the baseline period, the UCCs had 3,103 patient encounters with the ICD9 codes listed in Table 1, compared with 2,189 encounters during our intervention. There were no significant differences in the demographic variables of age or gender between periods (Table 2). The mean age of patients included for baseline data collection was 33.6 years, compared with a mean age of 35.5 years for the intervention period. Significant differences were noted in the distribution of diagnoses, provider type, and smoking status between the time periods.
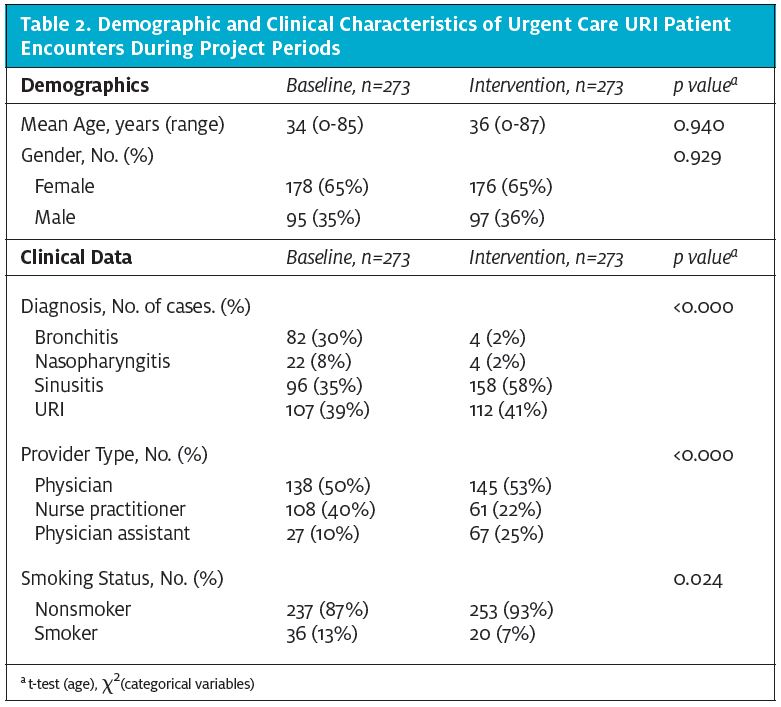
Suspected viral cases accounted for the majority of baseline encounters (n=211, nasopharyngitis, bronchitis and URI-NOS), but not during the intervention period (n=120). The majority of patients were nonsmokers; however, there were more smokers at baseline (13%) than during the intervention (7%). Physicians were the most frequent provider of care regardless of time period (50% at baseline vs 53% for intervention). However, more NPs provided care to patients than PAs at baseline than during the intervention period (40% vs 25%).
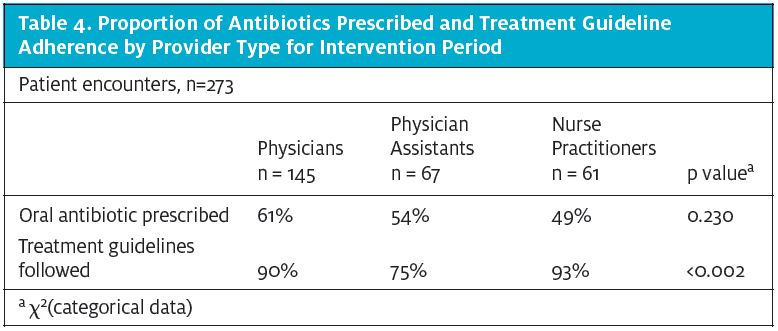
As a result of this project, the rate of antibiotic prescribing for the UCCs was reduced by 23.6% for bronchitis, sinusitis, nasopharyngitis, and URI-NOS visits collectively. There was a statistically significant change (p<0.05) in the proportion of patients prescribed an antibiotic as a result of the intervention (155/273, or 56.8%) compared with the baseline period (203/273, or 74.4%). After adjusting these results for diagnosis type, provider status, and smoking status, changes in the rate of antibiotic prescribing between our baseline and intervention groups remained significant (p<.000). For each additional patient receiving care for their URI during the intervention period, the odds of them receiving an antibiotic was reduced by a factor of .119, controlling for all other factors in the model (95% CI .060 to .237). The largest decrease in prescriptions were noted in the URI category, which demonstrated a 71% reduction from baseline (47%) to intervention (14%) as shown in Table 3. Smoking history was noted to be an independent predictor of antibiotic prescribing during the baseline period (p<.000), with 83% of smokers receiving an antibiotic for their URI. Furthermore, there was a 22% decrease in antibiotic prescriptions noted among smokers, with no significant differences (p=.441) noted between the smoking groups. Physicians prescribed a higher percentage of antibiotics than their PA and NP colleagues (61%, 54%, and 49%, respectively, p<0.230); however, the differences were not significant (Table 4.)
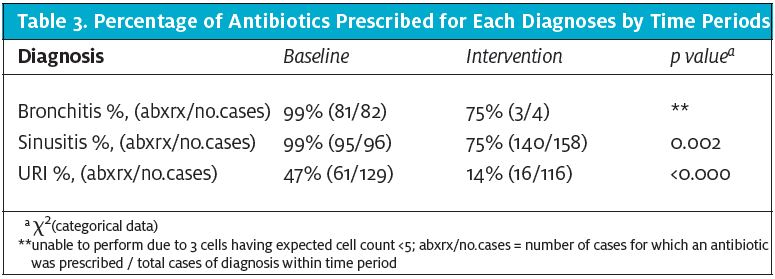
We obtained an 87% URI guideline compliance rate among providers during the intervention period. NPs were more likely to adhere to the guidelines, followed by physicians. The PA group had the lowest rate of adherence (p<0.002) (Table 4).
Using logistic regression, the physician and PA groups had a major influence on treatment guideline adherence. Treatment guidelines were less likely to be followed if a PA was the provider of care compared with the physician reference group with statistical significance—OR (95% CI) = .314 (.144, .685).
Discussion
Antibiotic overuse is a prevailing concern across the globe, as common infections present treatment challenges for healthcare providers and patients due to increasing antimicrobial resistance. To facilitate the judicious use of these drugs when treating common URI complaints in our setting, evidence-based interventions were reviewed for modeling and applicability. Most of the studies reviewed were conducted in the primary care setting.
However, an intervention specific to the unique characteristics and culture of the urgent care setting was desired. Our purpose was to determine if EBP interventions that have been successful in reducing in antibiotic prescribing and improving adherence to EBP guidelines with statistical significance could be effectively translated into the UC setting and provide similar results.
Utilizing a multimodal intervention inclusive of provider consensus meetings, EBP guideline review and adaptation, clinical pathways, prescriber feedback and audit, and patient education (inclusive of a marketing campaign), we were able to appreciate a modest, statistically significant decrease in the rate of antibiotic prescribing among the UC providers. Buy-in from providers was evidenced through an 87% rate of compliance with EBP guidelines. Individual components of the intervention were not tested for significance; however, providers agreed that each component contributed to the project’s success and their personal rate of adherence.
Similar to prior studies, we demonstrated a decrease in the proportion of antibiotics prescribed for patients with URIs with statistical significance.12,13,19,20 We reduced the rate of antibiotic use in sinusitis. However, prior studies measured broad-spectrum antibiotic use for this diagnosis as opposed to overall prescribing rate.8,9,21 We found only one study that targeted UCCs.18 Comparable to our results, in this study, NPs were more likely to comply with guidelines. Additionally, we noted the PA group had the lowest rate of guideline compliance. However, it is important for us to consider that 80% of the PAs were part-time providers. Only one of the five PAs had 100% participation at the consensus meetings, while the physicians and NPs had a complete 100% rate of attendance. Participation, rather than provider type, may be the stronger factor.
The full involvement of stakeholders most likely contributed to the success of our interventions, as in prior studies.13,22 We involved the providers whose antibiotic prescribing habits were being targeted, and provided them with compelling evidence that a change in practice was warranted. Patient education materials served as a complement to provider decision making and offered a credible source to guide care and facilitate compliance.
Conclusion
Antibiotic prescribing for common URI diagnoses was significantly reduced at two urgent care centers through the implementation of multimodal strategies targeting providers and patients. Such an approach could enhance compliance with URI evidence-based practice guidelines in the outpatient setting resulting in the judicious use of antibiotics.
Limitations
There are several limitations to this study. The first was the composition of the provider staff. Our provider groups were small and there was a change in providers between the baseline and intervention periods. Additionally, all but one PA was employed on a part-time basis. The baseline period included two providers not employed with the practice during the intervention period, and two of the physicians began with the practice just prior to the intervention. Therefore, prescribing patterns and guideline adherence by provider groups should be interpreted with caution. Changes in prescribing habits could be attributed to the intervention or a change in providers.
The second limitation is the possibility of the Hawthorne effect among providers. Providers could select another diagnosis to justify their choice to prescribe an antibiotic, thus improving their rate of guideline adherence for the audit.
Thirdly, it is difficult to determine if the prescriber feedback and audit was effective. Due to delays in the IRB approval process, the feedback and audit component of the intervention did not begin at the onset of the intervention. Feedback and audit was only provided for the 2014 months of October, November, and December at 3-week intervals. Similar to other studies, future replications of this intervention should allow for timely feedback to providers at regular intervals.8,13 Finally, we did not assess patient satisfaction prior to, during, or upon completion of this project. Capturing patient perception, acceptance, and adoption of the initiatives surrounding judicious antibiotic use in the urgent care setting would be essential for the expansion and sustainability of the program. The ability to demonstrate an increase or no change in patient satisfaction could assist in obtaining buy-in and adoption from additional urgent care owners and companies.
While multimodal interventions have demonstrated the most success in reducing antibiotic prescribing for URIs, we are unable to determine the individual benefits of each component. In the future, a confidential Likert-scale survey could be used to assess provider attitudes toward the individual components of the project and their effect on changing their prescribing habits.
Components from interventions used in ambulatory care settings were successfully translated into our UCCs to standardize care and reduce rates of antibiotic prescribing among providers treating URIs. With increasing rates of antimicrobial resistance globally, all providers should promote the judicious use of antibiotics. This project demonstrates through the involvement of frontline providers and patients, that practice variations can be reduced when EBP is fostered. Future studies should focus on the project’s sustainability, and whether this intervention would demonstrate similar results in other urgent care settings.
MELISSA JONES-HOLLEY, DNP, MSN, APRN, FNP-C and TENER VEENEMA GOODWIN, PHD, MPH, MS, RN, CPNP, FAAN
Citation: Jones-Holley M, Goodwin TV. A multimodal intervention to reduce antibiotic use for common upper respiratory infections in the urgent care setting. J Urgent Care Med. May 2017. Available at: https://www.jucm.com/multimodal-intervention-reduce-antibiotic-use-common-upper-respiratory-infections-urgent-care-setting/.
References
- Essack S, Pignatar AC. A framework for the non-antibiotic management of upper respiratory tract infections: towards a global change in antibiotic resistance. Int J Clin Pract Suppl. 2013 Nov;(180)4-9.
- Outterson K, Powers JH, Daniel GW, McClellan MB. Repairing the broken market for antibiotic innovation. Health Affairs. 2015;34(2):277-285.
- Arias CA, Murray BE. A new antibiotic and the evolution of resistance. N Engl J Med. 372(12):1168-1170.
- Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States, 2013. Washington, DC. 2013.
- Executive Order 13676: Combating antibiotic-resistant bacteria, 56931-5 (2014).
- Urgent Care Association of America. The Case for Urgent Care. Naperville, IL. 2011.
- Infectious Disease Society of America. Antibiotics drastically overprescribed for sore throats, bronchitis. 2013
- Gerber JS, Prasad PA, Fiks AG, et al. Effect of an outpatient antimicrobial stewardship intervention on broad-spectrum antibiotic prescribing by primary care pediatricians: a randomized trial. JAMA. 2013;309(22):2345-2352.
- Litvin CB, Ornstein SM, Wessell AM, et al. Use of an electronic health record clinical decision support tool to improve antibiotic prescribing for acute respiratory infections: the ABX-TRIP study. J Gen Intern Med, 2013;28(6):810-816.
- Razon Y, Ashkenazi S, Cohen A, et al. Effect of educational intervention on antibiotic prescription practices for upper respiratory infections in children: a multicentre study. J Antimicrob Chemother. 2005;56(6):937-940.
- Gonzales R, Anderer T, McCulloch CE, et al. A cluster randomized trial of decision support strategies for reducing antibiotic use in acute bronchitis. JAMA Intern Med. 2013;173(4):267-273.
- Jenkins TC, Irwin A, Coombs L, et al. Effects of clinical pathways for common outpatient infections on antibiotic prescribing. Am J Med. 2013;126(4):327-335.
- Metlay JP, Camargo CA Jr, MacKenzie T, et al. Cluster-randomized trial to improve antibiotic use for adults with acute respiratory infections treated in emergency departments. Ann Emerg Med. 2007;50(3):221-230.
- American Academy of Family Physicians. Leawood, KS.
- Gonzales R, Corbett KK, Leeman-Castillo BA, et al. The “minimizing antibiotic resistance in colorado” project: impact of patient education in improving antibiotic use in private office practices. Health Serv Res. 2005;40(1):101-116.
- van Driel ML, Coenen S, Dirven K, et al. What is the role of quality circles in strategies to optimise antibiotic prescribing? A pragmatic cluster-randomised controlled trial in primary care. Quality & Safety in Health Care. 2007;16(3):197-202.
- Every NR, Hochman J, Becker R, et al. Critical pathways: a review. Circulation. 2000;101(4):461-465.
- Harris RH, MacKenzie TD, Leeman-Castillo B, et al. Optimizing antibiotic prescribing for acute respiratory tract infections in an urban urgent care clinic. J Gen Intern Med. 2003;185(5):326-334.
- Juzych NS, Banerjee M, Essenmacher L, Lerner SA. Improvements in antimicrobial prescribing for treatment of upper respiratory tract infections through provider education. J Gen Intern Med. 2005;20(10):901-905.
- Welschen I, Kuyvenhoven MM, Hoes AW, Verheij TJ. Effectiveness of a multiple intervention to reduce antibiotic prescribing for respiratory tract symptoms in primary care: randomised controlled trial. BMJ. 2004;329(7463):431.
- Greene RA, Beckman H, Chamberlain J, et al. Increasing adherence to a community-based guideline for acute sinusitis through education, physician profiling, and financial incentives. Am J Man Care. 2004.10(10):670-678.
- Aagaard EM, Gonzales R, Camargo CA Jr, et al. Physician champions are key to improving antibiotic prescribing quality. Jt Comm J Qual Patient Saf. 2010;36(3):109-116.


