Published on
Urgent message: Polypharmacy—taking multiple medications to treat several chronic medical problems—puts patients at increased risk of developing additional health issues when they are prescribed even more medications for acute conditions in an urgent care center.
JASMEET SINGH BHOGAL, MD
It is not uncommon for patients, especially elderly patients, presenting to an urgent care center to have multiple medical problems. This makes it more likely that they are taking multiple medications as well. In fact, according to a 2010 report by the Centers for Disease Control and Prevention (CDC), almost 40% of older Americans have taken five or more prescription medications in a month. Twenty percent of Medicare beneficiaries have five or more chronic conditions, and 50% take five or more medications.1 The use of multiple medications presents a unique challenge to the urgent care provider: How do we balance the acute needs of the presenting condition with the potential for adverse reactions so commonly associated with polypharmacy?
This article focuses on the issues surrounding the use of multiple medications in an urgent care setting, with the goal of helping the urgent care provider make better care decisions while balancing the risk of adverse reactions with the benefit of treatment.
Polypharmacy
Polypharmacy is the use of multiple medications by a patient, including over-the-counter medications and herbal remedies.2 This is of particular concern in older patients, in whom there is already a greater risk of adverse events because of decreased medication clearance and other metabolic changes. The use of multiple medications is also independently associated with an increased risk for an adverse drug event (ADE), regardless of age.3 Prescribing in the face of polypharmacy requires significant caution, and the urgent care provider must seriously consider the implications of prescribing medications to any patient who is already taking multiple medications.
Two of the most important concerns associated with polypharmacy include the prescription of and use of inappropriate medications and the increased incidence of ADEs. Additionally, and perhaps underappreciated, the avoidance of medically necessary medication out of fear of adverse reactions is also common in the setting of polypharmacy. Each concern will be discussed here.
Inappropriate Medications
The prescription of inappropriate medications can have devastating implications for the patient in any setting, not just in an urgent care setting. Several tools have been developed to minimize the risk of inappropriate prescribing, including the Beers criteria, Screening Tool of Older Person’s Prescriptions (STOPP), and Fit fOR The Aged (FORTA). Of these, the Beers criteria are the most widely used.
The Beers Criteria
The Beers criteria are intended for use in ambulatory and institutional settings in patients aged 65 and older, with the goal of improving care by reducing patients’ exposure to potentially inappropriate medications (PIMs) and thus avoid ADEs. Established in 1991, the Beers criteria have undergone three revisions, most recently in 2012. The criteria are to be used along with clinical judgment. They are not intended to forbid the necessary use of the listed medications. When any of the medications are required, their use should be individualized for the patient, preferably by a medical team, to take into account all risks and benefits. When alternative medications cannot be found or used, patients taking them must be monitored for ADEs. The criteria include 53 medications categorized into three classes:
- Drugs that should be avoided
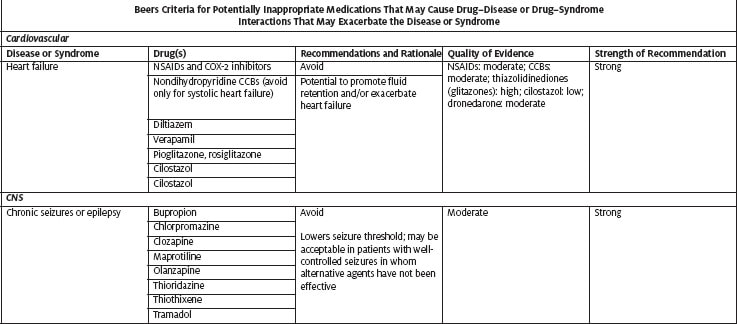
- Medications that are potentially inappropriate for use in older adults because of drug–disease or drug–syndrome interactions that may exacerbate the disease or syndrome
- PIMs to be used with caution in older adults
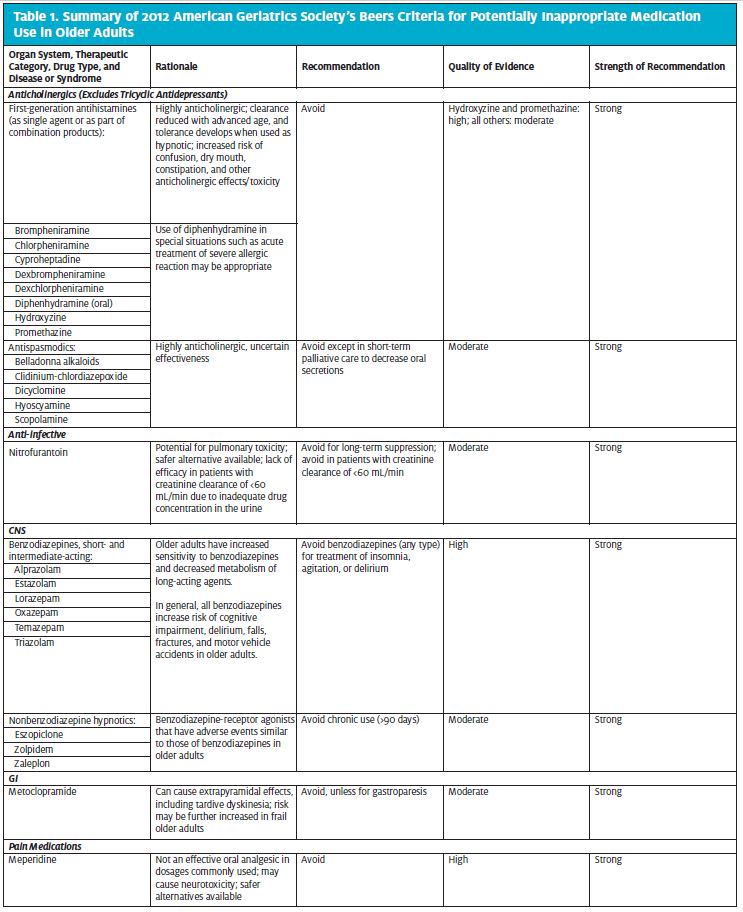
Table 1 highlights medications included in the 2012 American Geriatrics Society Beers criteria as PIMs for older adults. The table includes medication categories and individual medications that are commonly used in the urgent care setting and hence should be considered when treating older patients in that setting. The complete Beers lists are available on the following websites:
- https://www.americangeriatrics.org/health_care_professionals/clinical_practice/clinical_guidelines_recommendations/2012
- https://www.americangeriatrics.org/files/documents/beers/PrintableBeersPocketCard.pdf
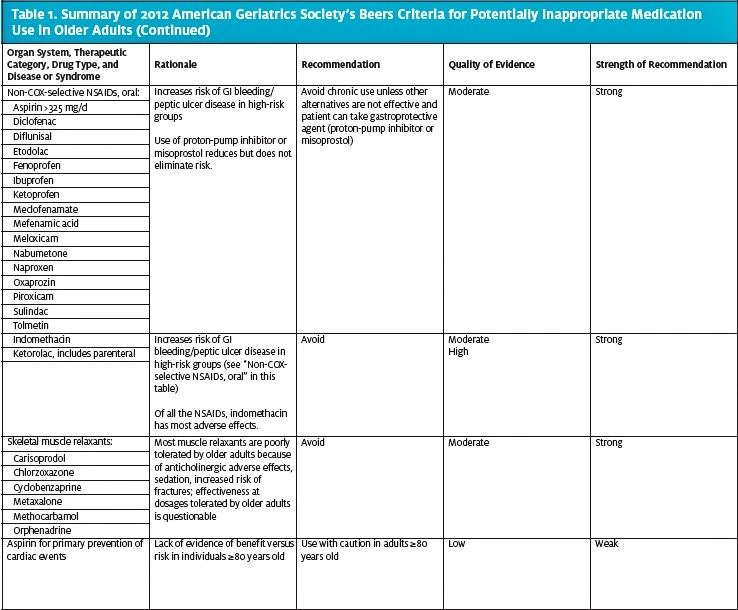
Of all the medications listed in the Beers criteria, anticholinergics deserve special attention because of their frequent use, and misuse, in the urgent care setting.
Anticholinergics
More than 1 in 5 elderly patients with dementia take drugs that have clinically significant anticholinergic activity.4 The use of anticholinergics in the general population is likewise commonplace and includes several over-the-counter medications. Adverse effects of these medications include the following:
- Confusion
- Hallucinations
- Dry mouth
- Blurred vision
- Constipation
- Nausea
- Urinary retention (precipitation of acute retention in existing benign prostatic hypertrophy)
- Impaired sweating
- Tachycardia
- Precipitation of acute glaucoma episode in existing narrow angle glaucoma
Although many medications have anticholinergic activity, some are routinely prescribed in the urgent care setting and therefore deserve special attention, including these:
- Antihistamines
- Cimetidine
- Cetirizine
- Chlorpheniramine
- Diphenhydramine
- Hydroxyzine
- Loratadine
- Ranitidine
- Antispasmodics
- Carisoprodol
- Dicyclomine
- Baclofen
- Antinausea and antidiarrhea agents
- Promethazine
- Loperamide
- Metoclopramide
The Anticholinergic Risk Scale has been developed to assess the risk of anticholinergic adverse effects.5 According to this scale, the risk of anticholinergic adverse effects increases if the score is 3 or higher. This scale can be used as a tool to identify patients who are at a risk of developing anticholinergic adverse effects when prescribed such medications. There is some evidence to show that this score may be useful in acute settings to improve risk stratification.6
In an urgent care setting, it can be helpful determining the pros and cons of prescribing these medications, especially in patients who are already taking medications that have anticholinergic effects. The cumulative effect of multiple medications on the list significantly adds to the risk of adverse events.
Adverse Drug Events
An ADE is defined as any injury that occurs from a drug, including noxious responses, drug administration errors, and any other circumstances that lead to an injury. Drug-related hospitalizations account for 2.4% to 6.5% of all medical admissions in the general population; the proportion is much higher for older patients.7 According to the CDC, more than 700,000 individuals are seen in hospital EDs for ADEs each year in the United States. Nearly 120,000 of these patients need to be hospitalized for further treatment. Older adults (aged 65 years or older) are also twice as likely as others to go to US EDs for ADEs, representing more than 177,000 emergency visits each year.
Factors that increase the risk of ADEs include
- Coexisting and multiple medical conditions
- Old age
- Use of multiple medication
Although it may be hard to avoid an ADE when a medication is being prescribed for the first time, certain scenarios must be investigated when prescribing new medications:
- Dose-related ADEs
- Drug–drug interactions
- Prescribing cascades
Dose-Related Adverse Drug Events
Dose-related ADEs are seen more commonly in failure to adjust drug doses for renal insufficiency or impairment. A common error is the failure to obtain a medical history of existing renal problems, or worse yet, obtaining such a history but not accounting for it when prescribing medications. Given the pace of urgent care work, this vital information can easily be missed. It is also important to recognize impairment in elderly patients, because advancing age itself leads to decreased renal function. Also, in the elderly population, the calculated creatinine clearance rate may not adequately reflect renal function.8 The commonly used methods used for determining creatinine clearance include the following:
- Cockcroft-Gault equation
- Modification of Diet in Renal Disease (MDRD) Study equation9
- Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation10
The Cockcroft-Gault equation is used to calculate creatinine clearance in patients with a stable serum creatinine level. It uses the patient’s sex, age, and lean body weight to calculate creatinine clearance. Although this equation was developed when obesity was less common, most drug-dosing guidelines have been developed using the Cockcroft-Gault equation. Given this, the most common drugs prescribed in the urgent care setting would require renal dose adjustments based on creatinine clearance, as derived from the Cockcroft-Gault equation. The equation, which does not use body surface area in calculating creatinine clearance, is as follows:
creatinine clearance = sex ×
[(140 – age)/(serum creatinine)] × (weight/72)
Dosing guidelines for patients who have decreased creatinine clearance are available via many resources, including medical software and websites such as Epocrates.com, Lexicomp Online (https://online.lexi.com), and Drugs.com (at http://www.drugs.com/dosage/). These are especially useful for safe dosing practices in elderly patients who require antibiotics to treat such conditions as pneumonia, urinary tract infections, and cellulitis.
Drug–Drug Interactions
Drug–drug interactions must be considered for every urgent care patient who is taking multiple medications. Risk of an ADE because of drug–drug interactions is substantially increased when multiple drugs are taken.11 Older patients are more prone than other groups to drug–drug interactions because they take more medications, including cardiac and blood-thinning medications, which have serious interactions with various other medications. Common examples of medications causing drug–drug interactions include the following:
- Nonsteroidal anti-inflammatory drugs (NSAIDs): When patients are receiving warfarin therapy, they are at increased risk of bleeding if they are given NSAIDs.
- Clarithromycin: Patients taking digoxin are 12 times more likely to develop digoxin toxicity if they are given clarithromycin.
- Azithromycin: According to warnings from the U.S. Food and Drug Administration, azithromycin can cause abnormal changes in the electrical activity of the heart that may lead to a potentially fatal irregular heart rhythm. Patients at particular risk for developing this condition include those with known risk factors, such as the following:
- Existing QT interval prolongation
- Low blood levels of potassium or magnesium
- Bradycardia
- Use of certain drugs used to treat arrhythmias
- Increased risk of QTc prolongation with use of azithromycin and citalopram, because both medications independently increase the risk of QTc prolongation
- Trimethoprim-sulfamethoxazole
- Increased risk of hypoglycemia when used with sulfonylureas
- Increased risk of hyperkalemia when used with
- Angiotensin-converting-enzyme inhibitors
- Angiotensin II receptor blockers
- Spironolactone
- Increased risk of serotonin syndrome with a combination of any of the following drugs:
- Amphetamines
- Cocaine
- Levodopa, carbidopa-levodopa
- Tramadol
- Selective serotonin reuptake inhibitors
- Bupropion
- Tricyclic antidepressants
- Ondansetron
- Valproate
- Carbamazepine
- Cyclobenzaprine
- Buspirone
- Triptans
- Ergot derivatives
- Lithium
| Table 2. Characteristics of the Main Types of Adverse Drug Reactions | |
| Type A | Type B |
| 85%–90% of all drug reactions | 10%–15% of all drug reactions |
| Occur in healthy individuals when given a sufficient dose and duration of the medication | Occur in a subgroup of patients who are susceptible |
| Predictable, based on known pharmacologic properties of the drug | Mostly unpredictable, usually mediated by immunologic response |
| Includes: | Includes: |
| Overdose | Idiosyncratic (not attributable to known pharmacologic properties of the drug; not immune-mediated either) |
| Adverse effects | Intolerance (adverse effects at subtherapeutic doses) |
| Drug interactions | Immunologic reaction (allergy) |
| Secondary effects | |
Urgent care providers should be actively looking for these interactions before prescribing medications. A thorough and systematic review of the patient’s medical history and the medications they are taking is very important to make sure that important drug–drug interactions are not missed when adding new medications at the urgent care visit. Also, if the medication has to be prescribed out of clinical necessity, a thorough discussion of drug interactions with the patient should be part of the overall discussion. This helps the patient understand what signs and symptoms to look for if an ADE does occur because of drug–drug interactions. Documentation of the following is necessary when prescribing despite the possibility of drug–drug interactions:
- Clinical decision making related to why the benefit outweighs the risk (low likelihood or low risk of interaction, no alternative, risk of nontreatment outweighs risk of adverse reaction)
- The patient’s understanding and acceptance of risk
- Anticipatory guidance for the patient
Various online resources Epocrates.com, Lexicomp Online (https://online.lexi.com), and Medscape’s Drug Interaction Checker (http://reference.medscape.com/drug- interactionchecker) are available for checking these interactions.
Prescribing Cascades
Prescribing cascades occur when a new drug is prescribed to treat symptoms arising from an unrecognized ADE related to an existing therapy.12 Again, this is of special concern in the elderly population. Given the fastpaced environment of the urgent care setting, these cascades can easily be overlooked without a dedicated effort to consider them. This is exacerbated when providers use more medications to treat the very symptoms created by existing medications. Sometimes, however, the initial treatment is unavoidable, and new medications have to be prescribed to control their adverse effects. Even when this is necessary, caution must be exercised to avoid further complications or adverse reactions. Examples of prescribing cascades include the following:
- Thiazide diuretic → gout → gout treatment
- NSAID → elevated blood pressure → anti-hypertension therapy
- Opioids → constipation → laxatives
- Antipsychotic medications → dystonia → diphenhydramine
Underutilization of Medications
In most circumstances, when clinically indicated medications are consciously avoided, the provider is attempting to do one of the following:
- Support compliance with essential medications
- Limit drug interactions
- Treat only active conditions
- Control the cost of treatment
An example of underutilization in the urgent care setting could include the avoidance of NSAIDs or oral steroids in patients presenting with an acute gout attack just because they have a history of diabetes or hypertension.
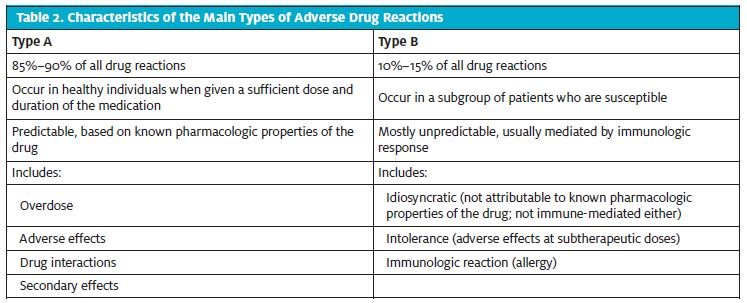
Drug Allergies
Drug allergies can be described as reactions that are produced as a result of an immune response to a medication. Adverse drug reactions can broadly be categorized into type A and type B (Table 2). Type A drug reactions include reactions secondary to drug overdose, adverse effects at therapeutic doses, drug interactions, and secondary effects of drugs. Type B reactions, on the other hand, are either immunologic reactions, adverse effects at sub-therapeutic levels, or reactions that are not attributable to the pharmacologic properties of the drug. Within Type B reactions, immunologic reactions can further be classified into the following types (Table 3):
- I: immediate reaction
- II: delayed reaction
- III: delayed reaction
- IV: delayed reaction
A description of the types of reactions is provided in Table 4. Although these form the majority of the immunologic responses that lead to drug eruptions, there are others.
Drug-Induced Autoimmunity
The pathogenesis of drug-induced autoimmunity is poorly understood. However, some drugs can induce autoimmune diseases. Examples of this reaction include immunoglobulin A (IgA) bullous dermatosis associated with ciprofloxacin, ceftriaxone, and metronidazole.
Fixed Drug Eruption
Fixed drug eruptions are relatively more common, although as is the case with drug-induced autoimmunity, the pathogenesis is also not clearly understood. The reaction is characterized by an erythematous and edematous plaque with a grayish center or central bullae. It commonly occurs on the lips, tongue, face, genitalia, and acral areas. Typical offending agents include sulfonamides, tetracyclines, NSAIDs, acetaminophen, and anticoagulants. Drug withdrawal and avoidance are the main treatment. Symptomatic treatment is also advised.
Pseudoallergic Reactions
Although some drug reactions appear to be related to immunity, they are not mediated by the immune system. These are called pseudoallergic reactions. The clinical presentation of these reactions is similar to a true allergic reaction, but they do not worsen with repeated exposure. Because the clinical presentation is the same, these reactions should be taken as seriously as any other IgE-mediated reaction and should be treated in the same manner. Also, because these reactions are not immunemediated, they are not diagnosed with skin testing or invitro allergy testing. The mechanism behind these reactions is not completely understood. However, they could be secondary to the following:
- Inhibition of prostaglandin production and increased leukotriene production
- Direct stimulation of mast cells, resulting in the release of mediators
- Vasovagal reflex
| Table 5. Examples of Pseudoallergic Reactions | |
| Drug | Clinical Reaction |
| Aspirin, nonsteroidal anti-inflammatory drugs | Asthma, exacerbation of rhinitis, urticaria/angioedema |
| Opioids | Pruritus, urticaria |
| Ciprofloxacin | Urticaria |
| Local anesthetics | Vasovagal syncope, anxiety |
Examples are provided in Table 5. NSAIDs are of particular importance here, considering their widespread use, their over-the-counter accessibility, and the sheer number of different reactions possible with this class. There are six types of allergic reactions to NSAIDs.13 Types 1 through 4 are recognized as pseudo allergic reactions, and types 5 and 6 are IgE-mediated allergic reactions. Type 5 and 6 reactions are usually caused by a single NSAID rather than
multiple NSAIDs. Pseudoallergic reactions, on the other hand, can be caused by any NSAID that is a cyclooxygenase-1 inhibitor. Type 5 and 6 reactions usually result from prior exposure and sensitization to the same agent,14 but that is not necessary for a pseudoallergic reaction to develop. The onset for type 5 and 6 reactions is usually minutes to a few hours after ingestion. For type 1, 2, 3, and 4 reactions, the onset is within 30 to 90 minutes. A description of the clinical features and associated comorbidities for each reaction type is provided in Table 6.
It is important to keep these reactions in mind when assessing patients with upper respiratory infections or cutaneous symptoms. Although these patients could have symptoms secondary to other etiologies, the use of NSAIDs could exaggerate existing problems.
Urgent care practitioners who are aware of the risk factors for drug allergies are much better able to avoid drug reactions in their patients.
Risk Factors for Drug Allergies
Important risk factors for developing drug allergies include the following:
- Female sex: For reasons not completely understood, women experience a higher frequency of both immediate and delayed drug reactions.
- Recurrent drug exposure: Repeated use of the same or related drugs is associated with higher rates of drug allergies.15
- History of allergic reactions to drugs: A history of allergic reactions to medications increases the risk that a patient will experience more drug reactions.
- Human leukocyte antigen (HLA) type: Patients’ HLA type is related to their familial propensity to develop immunologic drug reactions.
- Disease states: Examples include
- Atypical lymphocytes (e.g., Epstein-Barr virus, leukemia): reaction to aminopenicillins
- Acquired immunodeficiency syndrome: reaction to sulfonamides
Conclusion
As the complexity of health states increases in patients seen at urgent care centers, especially the elderly, so does the necessity of guarding against polypharmacy and other medication-related perils. In the fast-paced environment of an urgent care clinic, polypharmacy can be easily overlooked and lead to drug cascading, which can result in serious health issues. If these complications do occur, then urgent care providers must be able to recognize them and act on them appropriately. Likewise, prescription, over-the-counter, and herbal medications can all contribute to the development of significant and sometimes fatal adverse drug reactions and allergic reactions. Taking time to obtain a thorough medical history and medication history, especially in the elderly, can go a long way toward minimizing the risk of medicationrelated maladies in the urgent care setting.
References
- Tinetti ME, Bogardus ST Jr, Agostini JV. Potential pitfalls of disease-specific guidelines for patients with multiple conditions. N Engl J Med. 2004;351:2870–2874.
- Ferner RE, Aronson JK. Communicating information about drug safety. BMJ. 2006;333:143–145.
- Field TS, Gurwitz JH, Avorn J, et al. Risk factors for adverse drug events among nursing home residents. Arch Intern Med. 2001;161:1629–1634.
- Sura SD, Carnahan RM, Chen H, Aparasu RR. Prevalence and determinants of anticholinergic medication use in elderly dementia patients. Drugs Aging. 2013;30:837–844.
- Rudolph JL, Salow MJ, Angelini MC, et al. The anticholinergic risk scale and anticholinergic adverse effects in older persons. Arch Intern Med. 2008;168:508–513.
- Lowry E, Woodman RJ, Soiza RL, et al. Associations between the anticholinergic risk scale score and physical function: potential implications for adverse outcomes in older hospitalized patients. J Am Med Dir Assoc. 2011;2:565–572.
- Pirmohamed M, James S, Meakin S, et al. Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. BMJ. 2004;329:15–19.
- Garg AX, Papaioannou A, Ferko N, et al. Estimating the prevalence of renal insufficiency in seniors requiring long-term care. Kidney Int. 2004;65:649–653.
- Botev R, Mallié J-P, Couchoud C, et al. Estimating glomerular filtration rate: Cockcroft– Gault and Modification of Diet in Renal Disease formulas compared to renal inulin clearance. Clin J Am Soc Nephrol. 2009;4:899–906.
- Levey AS, Stevens LA, Schmid CH, et al; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612.
- Juurlink DN, Mamdani M, Kopp A, et al. Drug–drug interactions among elderly patients hospitalized for drug toxicity. JAMA. 2003;289:1652–1658.
- Rochon PA, Gurwitz JH. Optimising drug treatment for elderly people: the prescribing cascade. BMJ. 1997;315:1096–1099.
- Stevenson DD, Sanchez-Borges M, Szczeklik A. Classification of allergic and pseudoallergic reactions to drugs that inhibit cyclooxygenase enzymes. Ann Allergy Asthma Immunol. 2001;87:177–180.
- Quiralte J, Blanco C, Castillo R, et al. Anaphylactoid reactions due to nonsteroidal anti-inflammatory drugs: clinical and cross-reactivity studies. Ann Allergy Asthma Immunol.1997;78:293–296.
- Moss RB, Babin S, Hsu YP, et al. Allergy to semisynthetic penicillins in cystic fibrosis. J Pediatr. 1984;104:460–466.