Published on
Urgent Message: This retrospective chart review study found that the odds of children receiving an antibiotic prescription for acute otitis media in urgent care and the emergency department were significantly higher compared to a primary care setting. Further research is needed to better understand factors contributing to higher antibiotic prescribing rates in acute care settings.
Angelica M. Mangahas, BS; Heather M. Weinreich, MD, MPH; Johanna Wickemeyer, MD; Margaret Schmit MD; Rakhi Thambi, MD, MBA
Citation: Mangahas AM, Weinreich HM, Wickemeyer J, Schmit M, Thambi R. Influence of Clinical Setting on Antibiotic Prescribing Practices for Acute Otitis Media in Children at an Urban Academic Medical Center. J Urgent Care Med. 2024: 18(8):37-44
Abstract
Introduction: Acute otitis media (AOM) in children is a common cause of primary care and unscheduled visits and results in many antibiotic prescriptions. In most instances, the American Academy of Pediatrics’ (AAP) recommendations focus on “watchful waiting” with close follow-up. The AAP guidelines for the treatment of uncomplicated AOM only recommend initiation of antibiotic therapy in cases when there is no improvement within 48-72 hours. The purpose of this study was to investigate clinician prescribing practices at an urban academic hospital in accordance with the AAP guidelines in pediatric patients with uncomplicated AOM.
Methods: A retrospective, cross-sectional analysis of de-identified patient data was performed involving pediatric visits at the University of Illinois Hospital & Health Sciences System from January 2016 to May 2020. The primary outcome was treatment with an oral antibiotic for AOM. The secondary outcome was guideline concordance of antibiotic prescriptions. Descriptive statistics and chi-square analyses were performed. Logistic regression was used to predict antibiotic treatment and guideline adherence.
Results: In the study,792 patients met the inclusion criteria, and 85.7% received an oral antibiotic. Patients seen at an acute care facility—urgent care (UC) or the emergency department (ED)—were more likely to receive antibiotics compared to those seen in primary care clinics (64.7% vs. 33.0%, p<0.0001). The odds of receiving an antibiotic in an ED or UC setting were over 10 times greater than in a primary care clinic (OR 10.91 95% CI 5.67 to 20.98). If an antibiotic was prescribed, prescribing guidelines were followed 97.2% of the time. Deviations from antibiotic prescribing guidelines most commonly involved the use of macrolides.
Conclusion: Patients with AOM were more likely to receive antibiotic prescriptions in ED and UC settings compared to the primary care setting. When antibiotics are given, the appropriate first-line medications were prescribed the majority of the time.
Introduction
AOM is among the most common reasons for pediatric primary care visits in the United States. Approximately 80% of all children will have at least one episode of AOM in their lifetime, and one-third of children will have >5 episodes by the age of 7. AOM is most prevalent between 6 to 12 months of age, and the incidence declines after age 5 years.1
Most cases of AOM occur in the setting of viral respiratory infections and, in otherwise healthy children, will resolve without treatment. However, despite recommendations for delaying antibiotics in lower risk situations (eg, older children, unilateral symptoms, absence of fever) AOM remains among the most common indications for systemic antibiotic prescriptions in children.1,2
The American Academy of Pediatrics (AAP) recommends that mild to moderate AOM in patients older than 24 months be managed with either observation with close follow-up or oral antibiotic therapy based on shared decision-making with the patient’s parents/caregiver. Symptoms that categorize severe AOM include moderate or severe otalgia, otalgia for at least 48 hours, or a temperature of 39°C or higher.3 If antibiotics are clinically indicated, high-dose amoxicillin is the recommended initial treatment unless there is a known allergy. Cephalosporins are preferred and considered safe even if the patient is truly allergic to penicillin, and amoxicillin-clavulanate should be reserved for cases when the patient has taken amoxicillin in the past 30 days or has accompanying conjunctivitis.4
Adherence to guidelines among clinicians has remained imperfect, and the most recent evidence suggests that high rates of non-first-line antibiotic prescribing persists.5 The likelihood of adherence to antibiotic guidelines has also been shown to vary based on clinician specialty, age of the child, location of care, and time of the visit.6 Accurate diagnosis and appropriate treatment, specifically decreasing the use of immediate antibiotics to treat mild-moderate AOM, has important implications for antibiotic resistance.7 Few studies have investigated antibiotic prescribing practices in an ethnically diverse and low-income population. The objective of this study was to investigate clinician prescribing practices in a large, urban academic hospital system in accordance with the AAP clinical practice guidelines (CPG).
Methods
Ethical Considerations
The University of Illinois at Chicago Institutional Review Board approved the study protocol (#2019-1192). The content is solely the responsibility of the authors and does not necessarily represent the official views of the Center for Clinical and Translation Science (CCTS).
Study Design
This study was a retrospective analysis of an administrative dataset. Data for this study was extracted with the support of the CCTS using the Clinical Research Data Warehouse (CRDW) and the University of Illinois at Chicago Clinical Information Repository for Cohort Learning and Exploration (UIC CIRCLE) as the data source. The CRDW is a de-identified data store of 3.4 billion electronic health records, including 260 million clinical events and 6.2 million visit encounters within the University of Illinois Hospital & Health Sciences System (UI Health). The database includes information regarding the clinician, medications, billing codes, procedures, patient demographics, vital signs, labs, and diagnoses associated with various encounters at any of the UI health subsites. UI Health is a part of the University of Illinois at Chicago (UIC), which includes a 462-bed tertiary care hospital, 26 outpatient clinics, and 14 primary care facilities throughout the city of Chicago.
Patient Population
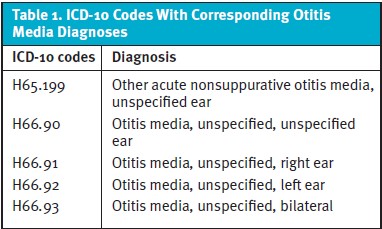
The study population consisted of all pediatric patients in the CRDW with the International Classification of Diseases-10 (ICD-10) codes for AOM assigned during an initial visit and subsequent ambulatory encounter within UI Health from January 1, 2016 – May 20, 2020. We restricted our analysis to 1,000 consecutive charts, which were collected by CCTS sequentially and blinded to the authors. Cases were not selected preferentially based on the location of treatment. (Table 1). Inclusion criteria consisted of patients aged 2 to 12 years with the corresponding ICD-10 codes during an initial visit during this time frame. The age range was chosen in correlation to the AAP guideline for non-severe AOM for older children (>24 months). Exclusion criteria included the following: patients older than 12 years of age, follow-up visits related to AOM, as well as inpatient admission or admission for surgical procedures (eg, tympanostomy tubes).
Medications prescribed at each encounter, location of the visit, clinician degrees (Doctor of Medicine [MD], Doctor of Osteopathic Medicine [DO], Advanced Practice Registered Nurse [APRN], which is similar to Nurse Practitioner [NP], and Physician Assistant [PA]), and demographic information of the patient including age, sex, race, and zip code were obtained from the CRDW.
Independent and Outcome Variables
The primary outcome was the proportion of patients who were treated with an oral antibiotic following a diagnosis of AOM. If a patient was not prescribed any antibiotics, then “watchful waiting” was assumed. Medications prescribed at the time of the encounter were collected and analyzed.
The secondary outcome was the proportion of patients who were treated with an oral antibiotic that followed the AAP CPGs if a prescription was provided.
Statistical Analysis
Percentages were calculated for categorical variables (eg, sex, race, location of visit, prescribing clinical credentials). Chi-square was performed to analyze percent differences between patients who received antibiotics versus no antibiotics. Univariate and multivariate logistic regression (age, sex, location, prescribing clinician, race) models were used to predict treatment with oral antibiotics versus no antibiotics. Patients aged 2-6, male sex, MD/DO, and primary care facilities were used for reference categories. A p-value of less than 0.05 was considered statistically significant. Statistical analyses were performed using MedCalc for Windows, version 19.4 (MedCalc Software, Ostend, Belgium).
Results
Demographics
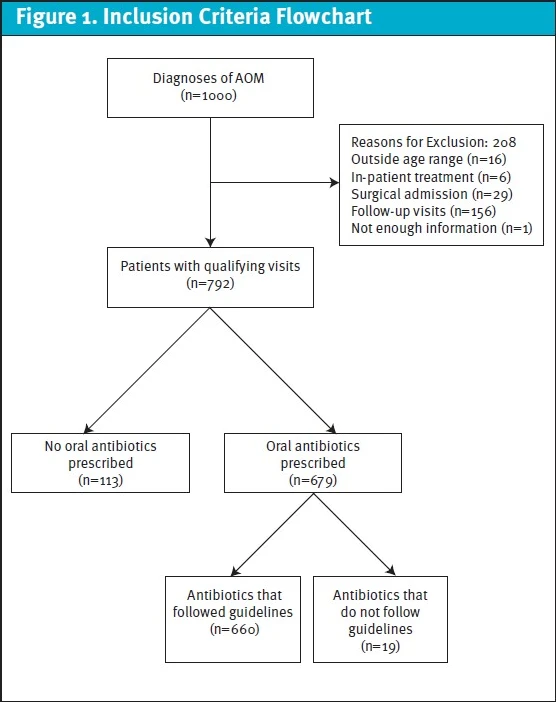
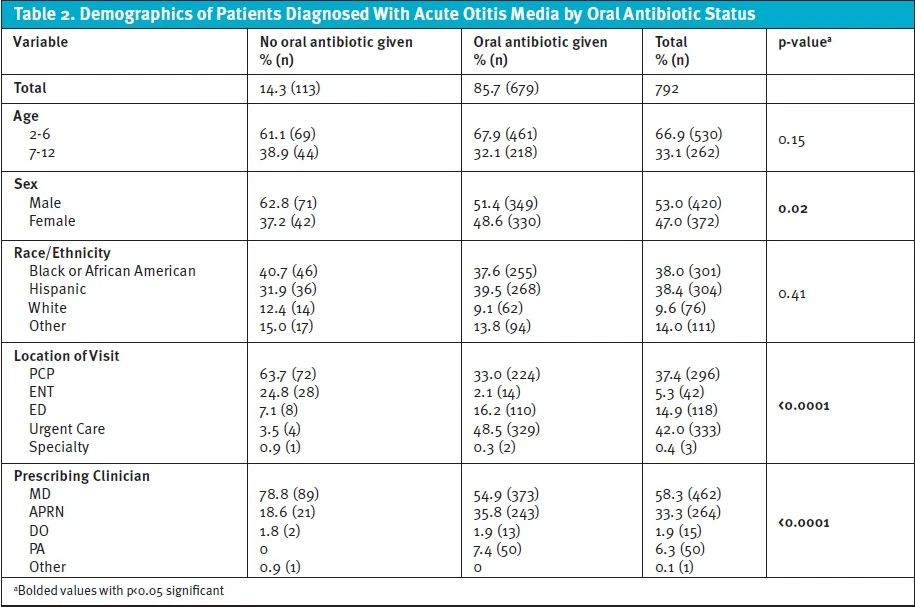
Of the 1,000 patient encounters that were extracted from the database, a total of 792 patients met inclusion criteria (Figure 1). The patient population consisted of a majority of children between the ages of 2 and 6 (66.9%), with 53.0% being male. A range of racial groups were represented including Hispanic (38.4%), African American (38.0%), White (9.6%), and Other (14.0%). Many patients were treated at a UC facility (42.0%), primary care facility (37.4%), and ED (14.9%), with a small percentage of patients at the otolaryngology clinic (5.3%) and at a specialty clinic (0.4%) Most patients were evaluated by an MD (58.3%) or an APRN (33.3%).
Primary Outcome (Treatment With Oral Antibiotics)
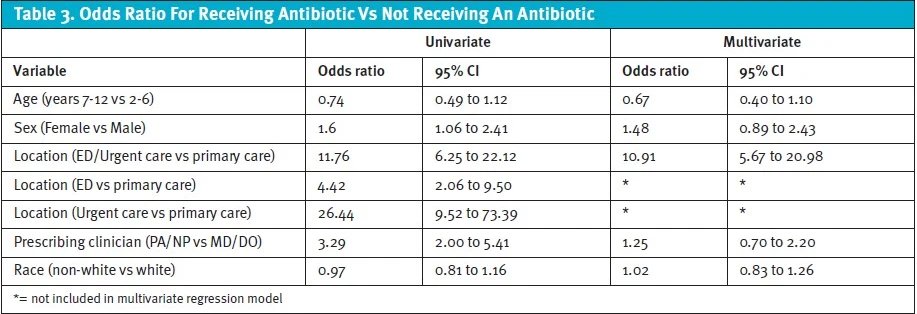
Of the 792 patients who presented with AOM and met criteria, 85.7% received an oral antibiotic. There were no differences between oral antibiotic prescriptions between children based on age and race (Table 2); however, males were more likely to receive an oral antibiotic compared to female patients (51.4% vs. 48.6%, p=0.02). There were significant differences in the odds of receiving an antibiotic prescription depending on the setting of care. Patients were more likely to receive an antibiotic in an ED or UC facility compared to primary care settings (64.7% vs. 33.0%, p<0.0001). Of the clinicians prescribing antibiotics, MDs made up the majority (54.9%), followed by APRNs (35.8%), and PAs (7.4%). However, APRNs and PAs were more likely to prescribe antibiotics compared to MDs (78.8% vs. 54.9% p<0.0001) (Table 2).
Following multivariate regression (age, sex, location, prescribing clinician, race), only the encounter location was significant. In comparison to a primary care facility, presentation to either the ED or UC conveyed an increased odds of 10.91 of receiving an antibiotic (OR 10.91, 95% CI 5.67 to 20.98). When separating acute care settings, univariate regression showed that, relative to primary care visits, presentation to an UC was associated with a greater odds of receiving an antibiotic compared to patients seen in the ED (UC OR 26.44, 95% CI 9.52 to 73.39 versus ED OR 4.42, 95% CI 2.06 to 9.50) (Table 3).
Secondary Outcomes (Antibiotic Prescription)
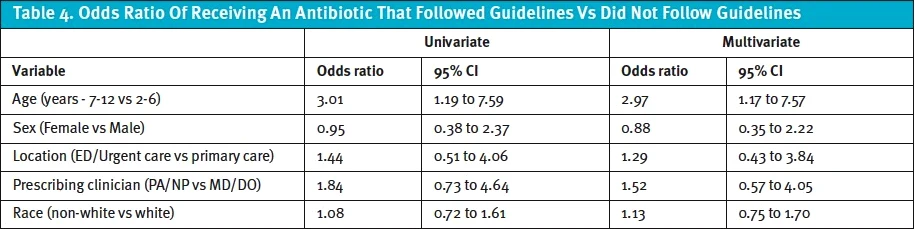
Of those who did receive an antibiotic (n=679), 97.2% received an antibiotic that followed prescription practices dictated by the CPG. Significantly more children 7-12 years of age compared to those ≤6 years of age received an antibiotic that did not follow guidelines (57.9% vs. 42.1%, p=0.01). There were no differences by gender, race, location of the visit, or credentials of prescribing clinicians. Following multivariate regression analysis (age, sex, location, prescribing clinician, race), the odds of being prescribed an antibiotic that did not follow guidelines was 2.97 for those aged 7 to 12 compared to patients ≤ 6 years of age (OR 2.97, 95% CI 1.17 to 7.57) (Table 4). Of those that received an antibiotic that did not follow guidelines (n=17), macrolides were the most common (88.2%) followed by clindamycin (5.9%), and trimethoprim-sulfamethoxazole (5.9%).
Other Medications
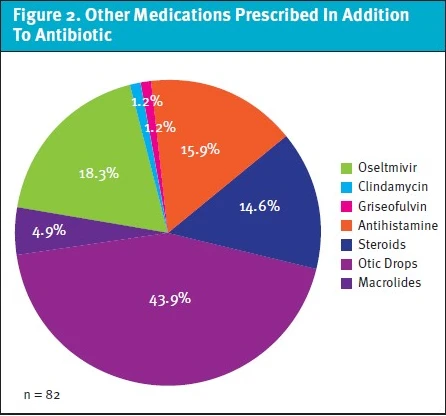
Eleven patients were prescribed non-antibiotic medications. These included:otic drops (antibacterial and/or steroid) (45.5%), oral steroids (36.4%), and oral/nasal antihistamines (18.2%). Of those who were prescribed medications in addition to an antibiotic (n=82), otic antibiotic drops (43.9%), and oseltamivir (18.3%) were the most common (Figure 2).
Discussion
Among a large, urban population of children presenting with AOM, 85.7% of patients in our sample were prescribed oral antibiotics at their initial encounter. The clinical setting in which patients were treated was most predictive variable among those examined for receiving an antibiotic prescription. Collectively, the ED and UC facilities were significantly more likely to prescribe oral antibiotics for AOM compared to primary care settings; however, in univariate analysis being seen in UC was associated with even higher odds of receiving an antibiotic than the ED. Of those who did receive an antibiotic prescription, the correct antibiotic was prescribed 97.2% of the time. Older children were more likely to receive non-first-line antibiotics.
Although the AAP guidelines recommend “watchful waiting” with close follow-up or antibiotic therapy treatment based on joint decision making, though “watchful waiting” with close follow-up would avoid many antibiotic prescriptions as most cases of AOM are viral.1 Additionally, in a study that evaluated 1,114 cases of AOM, only 35% of cases were classified as severe. Other studies have indicated a reduction in the incidence of severe AOM following the introduction of the pneumococcal conjugate vaccinations and the Hemophilus influenzae vaccinations.8,9 For this reason, it is increasingly likely that most cases of AOM do not meet criteria as “severe.” Our study demonstrated that antibiotic over-prescribing was common in our hospital system, especially in the acute care settings. Further studies are needed to understand the hesitant adoption of “watchful waiting” practices in the management of uncomplicated AOM at these clinical locations. “Watchful waiting” may hypothetically be avoided in these settings for many reasons: clinician concerns for lack of patient follow-up, inability to observe for complications, more severe presentations tending to present for unscheduled, acute care, among others. Another reason for high antibiotic prescription rates in acute care settings may stem from parental belief that antibiotic administration results in quicker resolution of symptoms as well as poor caregiver assessment of the risks and benefits of antibiotic administration in children with viral respiratory infections.10 This may be reflected by the higher numbers of older children (ie, >2 years) receiving antibiotics in comparison to other ages, as these children make up the population of those who attend daycare and kindergarten. Our study also found that <5% of patients among our sample who presented to UC did not receive an antibiotic prescription. Since UC clinicians care for patients they do not have a prior relationship with, children’s caregivers may be less inclined to leave without receiving an antibiotic prescription.11
Time pressures may also be a factor in why we observed higher rates of antibiotic prescribing in ED and UC centers. Both the ED and UC are fast-paced environments with high patient volumes. Frequently, clinicians in these settings commonly feel they do not have enough time to spend with each patient. Unsurprisingly, clinicians are more prone to inappropriate prescribing decisions with shorter visits. Conversely, the longer clinicians spend with patients, the less likely they are to prescribe antibiotics.12
In our study, prescriber credentials did not appear to be a predictor of the decision to prescribe antibiotics. Other studies conducted in both pediatric and adult populations have demonstrated non-physician prescribers were more likely to prescribe antibiotics, and rates of “watchful waiting” were lower among PAs and NPs than physicians.13,14 These differences could be due to varying levels of comfort and experience, but also due to patients and families being less inclined to accept recommendations from non-physician clinicians. A recent study from the University of Colorado demonstrated that a simple antimicrobial education course for prescribers decreased the frequency of prescribing antibiotics at the initial visit and increased the proportion of prescribers who employed “watchful waiting” techniques.15
It is encouraging that first-line antibiotics were selected in the vast majority of cases among the charts reviewed in our study. It is likely this is facilitated by the fact that antibiotic guidelines for pediatric AOM are straightforward and have not changed dramatically in recent years.16 There remained, however, a small subset of patients in our study who were treated with non-first-line antibiotics. Non-first-line medications are generally more expensive than amoxicillin, putting a greater strain on both state funded insurance and private insurance systems.17,18 We also observed macrolides were the most common inappropriate antibiotic prescribed. A recent meta-analysis suggested that when treating AOM, macrolides have a higher risk of clinical failures, which is believed to be attributable to increasing rates of S. pneumoniae resistance.19
Our study also found occasional prescribing of additional non-antibiotic medications for AOM. Medications such as antihistamines and nasal and oral steroids have a limited role in managing Eustachian tube dysfunction and have not been shown shorten the course of AOM. Furthermore, in cases where the tympanic membrane is intact and there is no evidence of otitis externa, otic antibiotic drops are not recommended.20-22 Antivirals for influenza have been shown to be associated with a slight reduction in associated AOM in younger children with symptomatic influenza infections.23 Our study showed that oseltamivir was prescribed in 18.3% of patients who received appropriate antibiotics, however, due to limitations of de-identified data and retrospective design, it is unclear if these patients also had influenza.
The “watchful waiting” approach for AOM centers around antibiotic stewardship. According to the Centers for Disease Control and Prevention (CDC), at least 30% of outpatient antibiotics prescribed in the United States are unnecessary.24 There are about 8.7 million antibiotic prescriptions written annually for AOM alone in the United States.5 Given how common the diagnosis of AOM is, it represents one of the larger opportunities for improved stewardship. Antibiotic over-prescribing is often associated with unknown etiology of infections.25 Furthermore, patients commonly present explicitly with requests for antibiotic prescriptions.25
A strength of this study is the large dataset from a diverse, urban setting from which the data were extracted. Consequently, however, the findings of our study would be most applicable to other large healthcare systems that serve low-income populations with high rates of ED utilization. Of all cases, the majority of cases of AOM in this study presented to the ED or UC (56.9%) compared to a national rate of 7.8% reported in a 2018 study. Low socioeconomic status, lack of access to primary care, lack of insurance coverage, and perceived urgency of conditions are among many reasons for increased ED usage within a hospital system.26 Most caregivers within our population have low education and health literacy, particularly about common childhood illnesses, such as otitis media. Being born outside the United States, of minority ethnicity or race, having low educational achievement, younger age, or having a child publicly insured are correlated with low health literacy, which leads to increased rates of emergency room visits, and greater odds of non-urgent cases.27 In one study with patient populations of low socioeconomic status in urban settings and publicly-run hospital systems, only 23.3% of pediatric patient caregivers attempted to contact their primary care office before going to the ED. Other caregivers who brought their child to the ED stated that they perceived lack of access to the primary care clinic, lack of knowledge of after-hour phone lines, and the belief that they would receive faster care in the ED.28

It is important to note the various limitations of this study. First and most importantly, this study was limited by retrospective design and the use of de-identified data, so sensitivity analyses were not feasible to determine the accuracy of statistical assumptions. Second, as data were extracted with the support of CCTS using the CRDW, clinically important information in the diagnosis of AOM, such as physical exam findings and vital signs, were unavailable. Instead, ICD-10 codes determined patient inclusion in our study. Therefore, it cannot be confirmed that patients who received otic drops did not have a tympanic perforation or myringotomy tubes, for example. Furthermore, patients’ symptoms were not described, so it cannot be confirmed that patients who received oral antibiotics did not have severe AOM. Another limitation of this study included the lack of confirmation if the prescriptions were filled by the caregivers or if the antibiotics were prescribed with the understanding that it would only be filled if the patient’s symptoms worsen after an appropriate period of observation, which may often have been the case. Therefore, it is likely that the prescription rates overestimated the actual antibiotic consumption rates, however, this would be expected to be similarly overestimated across settings. Additionally, location of care was determined based on the prescriber’s department. Thus, if a prescriber worked in multiple departments, it is possible that their department at the time of prescription did not accurately reflect the site of care. Another limitation was that our multivariate regression model found that, while age predicted the odds of being prescribed an inappropriate antibiotic, data on many other variables, such as insurance coverage, were unavailable. Finally, this data was collected during 2016-2020 prior to the COVID-19 pandemic. The number of UC centers has increased by more than 50% since 2016, and public use of UC has also increased dramatically during this time.29 Therefore, the decision of caregivers to seek care when their children have otalgia in UC vs ED vs primary care settings is likely markedly different compared to just a short time ago. For this reason, after the COVID-19 pandemic, it is likely that both clinician and patient opinions about antibiotic prescribing have changed. However, one recent, large, retrospective study of patients in Dubai showed ongoing over-prescribing of antibiotics post COVID-19.30 More studies are necessary to determine how caregivers and clinicians behave in terms of appropriate use of antibiotics for pediatric AOM in the wake of the many changes associated with COVID-19.
The AAP guidelines recommend “watchful waiting” with close follow-up or antibiotic therapy based on joint decision making for most children with AOM. This study is important in identifying which clinicians and locations may be more likely to deviate from these guidelines, especially in an urban setting with diverse patient populations. Future research would be of value to determine if the trends observed in our study population before COVID-19 persist, given the rapid changes in UC utilization and clinician credentials and changes among rural and suburban EDs and UC centers. Many organizations and governmental agencies, including CDC, the Urgent Care Association, and Infectious Disease Society of America, have created task forces and issued guidelines surrounding antibiotic stewardship in the outpatient setting in recent years.31 Importantly, to add to our work, prospective studies of treatment decisions for pediatric UC patients with AOM are needed to determine if efforts around antibiotic stewardship in the outpatient population are leading to improvements in guideline coherent prescribing and which additional areas represent the greatest opportunities to invest our continuing efforts around this important goal.
Disclaimer: The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Sources of Support: We acknowledge the University of Illinois Chicago Center for Clinical and Translational Science and their extraction of this data which was supported by the National Center for Advancing Translation Sciences, National Institutes of Health, through Grant Award Number UL1TR0020023.
Manuscript submitted December 8, 2023; accepted April 3, 2024
Author Affiliations: Angelica M. Mangahas, BS, University of Illinois. Heather M. Weinreich, MD, MPH, University of Illinois Health System. Johanna Wickemeyer, MD, Yale School of Medicine. Margaret Schmit MD, University of Wisconsin. Rakhi Thambi, MD, MBA, University of Illinois Health System.
References
- Schilder AG, Chonmaitree T, Cripps AW, et al. Otitis media. Nat Rev Dis Primers. Sep 8 2016;2(1):16063. doi:10.1038/nrdp.2016.63
- Schilder AG, Marom T, Bhutta MF, et al. Panel 7: Otitis Media: Treatment and Complications. Otolaryngol Head Neck Surg. Apr 2017;156(4_suppl):S88-s105. doi:10.1177/0194599816633697
- Lieberthal AS, Carroll AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. Mar 2013;131(3):e964-99. doi:10.1542/peds.2012-3488
- Harmes KM, Blackwood RA, Burrows HL, Cooke JM, Harrison RV, Passamani PP. Otitis media: diagnosis and treatment. Am Fam Physician. Oct 01 2013;88(7):435-40.
- Frost HM, Becker LF, Knepper BC, Shihadeh KC, Jenkins TC. Antibiotic Prescribing Patterns for Acute Otitis Media for Children 2 Years and Older. J Pediatr. May 2020;220:109-115.e1. doi:10.1016/j.jpeds.2020.01.045
- Shviro-Roseman N, Reuveni H, Gazala E, Leibovitz E. Adherence to acute otitis media treatment guidelines among primary health care providers in Israel. Braz J Infect Dis. 2014;18(4):355-9. doi:10.1016/j.bjid.2013.11.007
- Haggard M. Poor adherence to antibiotic prescribing guidelines in acute otitis media-obstacles, implications, and possible solutions. Eur J Pediatr. Mar 2011;170(3):323-32. doi:10.1007/s00431-010-1286-4
- Marra LP, Sartori AL, Martinez-Silveira MS, Toscano CM, Andrade AL. Effectiveness of Pneumococcal Vaccines on Otitis Media in Children: A Systematic Review. Value Health. Jun 2022;25(6):1042-1056. doi:10.1016/j.jval.2021.12.012
- Van Dyke MK, Pirçon JY, Cohen R, et al. Etiology of Acute Otitis Media in Children Less Than 5 Years of Age: A Pooled Analysis of 10 Similarly Designed Observational Studies. Pediatr Infect Dis J. Mar 2017;36(3):274-281. doi:10.1097/inf.0000000000001420
- Zahreddine L, Hallit S, Shakaroun S, Al-Hajje A, Awada S, Lahoud N. Knowledge of pharmacists and parents towards antibiotic use in pediatrics: a cross-sectional study in Lebanon. Pharm Pract (Granada). Jul-Sep 2018;16(3):1194. doi:10.18549/PharmPract.2018.03.1194
- Incze MA, Redberg RF, Katz MH. Overprescription in Urgent Care Clinics-The Fast and the Spurious. JAMA Intern Med. Sep 1 2018;178(9):1269-1270. doi:10.1001/jamainternmed.2018.1628
- Neprash HT, Mulcahy JF, Cross DA, Gaugler JE, Golberstein E, Ganguli I. Association of Primary Care Visit Length With Potentially Inappropriate Prescribing. JAMA Health Forum. Mar 3 2023;4(3):e230052. doi:10.1001/jamahealthforum.2023.0052
- Roumie CL, Halasa NB, Edwards KM, Zhu Y, Dittus RS, Griffin MR. Differences in antibiotic prescribing among physicians, residents, and nonphysician clinicians. Am J Med. Jun 2005;118(6):641-8. doi:10.1016/j.amjmed.2005.02.013
- Young EH, Panchal RM, Yap AG, Reveles KR. National Trends in Oral Antibiotic Prescribing in United States Physician Offices from 2009 to 2016. Pharmacotherapy. Oct 2020;40(10):1012-1021. doi:10.1002/phar.2456
- Frost HM, Monti JD, Andersen LM, et al. Improving Delayed Antibiotic Prescribing for Acute Otitis Media. Pediatrics. Jun 2021;147(6)doi:10.1542/peds.2020-026062
- Deniz Y, van Uum RT, de Hoog MLA, Schilder AGM, Damoiseaux R, Venekamp RP. Impact of acute otitis media clinical practice guidelines on antibiotic and analgesic prescriptions: a systematic review. Arch Dis Child. Jun 2018;103(6):597-602. doi:10.1136/archdischild-2017-314103
- Wattles BA, Smith MJ, Feygin Y, et al. Inappropriate Prescribing of Antibiotics to Pediatric Patients Receiving Medicaid: Comparison of High-Volume and Non-High-Volume Antibiotic Prescribers-Kentucky, 2019. Healthcare (Basel). Aug 16 2023;11(16)doi:10.3390/healthcare11162307
- Chua KP, Fischer MA, Linder JA. Appropriateness of outpatient antibiotic prescribing among privately insured US patients: ICD-10-CM based cross sectional study. BMJ. Jan 16 2019;364:k5092. doi:10.1136/bmj.k5092
- Frost HM, Bizune D, Gerber JS, Hersh AL, Hicks LA, Tsay SV. Amoxicillin Versus Other Antibiotic Agents for the Treatment of Acute Otitis Media in Children. J Pediatr. Dec 2022;251:98-104.e5. doi:10.1016/j.jpeds.2022.07.053
- Rosenfeld RM, Schwartz SR, Cannon CR, et al. Clinical practice guideline: acute otitis externa executive summary. Otolaryngol Head Neck Surg. 2014;150(2):161-168. doi:10.1177/0194599813517659
- Rosenfeld RM, Tunkel DE, Schwartz SR, et al. Clinical Practice Guideline: Tympanostomy Tubes in Children (Update). Otolaryngol Head Neck Surg. Feb 2022;166(1_suppl):S1-s55. doi:10.1177/01945998211065662
- Lou Z, Lou Z, Tang Y, Xiao J. The effect of ofloxacin otic drops on the regeneration of human traumatic tympanic membrane perforations. Clin Otolaryngol. 2016;41(5):564-570. doi:10.1111/coa.12564
- Winther B, Block SL, Reisinger K, Dutkowski R. Impact of oseltamivir treatment on the incidence and course of acute otitis media in children with influenza. Int J Pediatr
- Measuring Outpatient Antibiotic Prescribing. Centers for Disease Control and Prevention. Accessed July 1, 2023. https://www.cdc.gov/antibiotic-use/data/outpatient-prescribing/index.html#f2
- Otaigbe, II, Elikwu CJ. Drivers of inappropriate antibiotic use in low- and middle-income countries. JAC Antimicrob Resist. Jun 2023;5(3):dlad062. doi:10.1093/jacamr/dlad062
Otorhinolaryngol. Jun 2010;74(6):684-8. doi:10.1016/j.ijporl.2010.03.024 - Tong S, Amand C, Kieffer A, Kyaw MH. Trends in healthcare utilization and costs associated with acute otitis media in the United States during 2008-2014. BMC Health Serv Res. May 02 2018;18(1):318. doi:10.1186/s12913-018-3139-1
- May M, Brousseau DC, Nelson DA, et al. Why Parents Seek Care for Acute Illness in the Clinic or the ED: The Role of Health Literacy. Acad Pediatr. Apr 2018;18(3):289-296. doi:10.1016/j.acap.2017.06.010
- Pethe K, Baxterbeck A, Rosenthal SL, Stockwell MS. Why Parents Use the Emergency Department Despite Having a Medical Home. Clin Pediatr (Phila). Jan 2019;58(1):95-99. doi:10.1177/0009922818809447
- Urgent Care Association. Urgent Care Industry White Paper: The Essential Nature of Urgent Care in the Healthcare Ecosystem Post-COVID-19. August 17, 2023.
- Moradigaravand D, Senok A, Al-Dabal L, et al. Unveiling the dynamics of antimicrobial utilization and resistance in a large hospital network over five years: Insights from health record data analysis. PLOS Digit Health. Dec 2023;2(12):e0000424. doi:10.1371/journal.pdig.0000424
- Amin AN, Dellinger EP, Harnett G, et al. It’s about the patients: Practical antibiotic stewardship in outpatient settings in the United States. Front Med (Lausanne). 2022;9:901980. doi:10.3389/fmed.2022.901980
Download the article PDF: Influence of Clinical Setting on Antibiotic Prescribing Practices for Acute Otitis Media in Children at an Urban Academic Medical Center
