Published on
Urgent message: Not all patients presenting to urgent care with chest pain require immediate transfer to a higher-acuity setting. Rather, immediate evaluation in the urgent care center can inform next steps for management on site or, in the event of truly emergent symptoms, transfer.
Tushar Menon, MD; Sahil Kapur, DO; Ameera C. Mistry, MD; and David M. Boyd, MD
Key words: T-call lymphoblastic lymphoma, non-Hodgkin’s lymphoma, anterior mediastinal mass, chest pain
ABSTRACT
Chest pain patients require timely evaluation due to the broad differentials of diagnosis including acute coronary syndrome (ACS), aortic dissection, myocarditis, pulmonary embolism (PE), tension pneumothorax , gastroesophageal reflux disease (GERD), or musculoskeletal pain. Sometimes, a less common diagnosis such as malignancy is found.
INTRODUCTION
T-cell lymphoblastic lymphoma (T-LBL) is a rare form of aggressive non-Hodgkin’s lymphoma that comprises about 2% of all non-Hodgkin’s lymphomas.1 T-LBL itself accounts for 90% of all LBLs and generally occurs in adolescents and young adults, mostly males.2 They typically present as an advanced, widely disseminated version of non-Hodgkin’s lymphoma, usually with a mediastinal bulky mass with concomitant pleural and pericardial effusions. Typically, the thymus, testes, and ovaries are involved.3 CNS involvement can be seen in 5% to 10% of cases.4
CASE PRESENTATION
A 20-year-old male with no significant past medical history presented with a 3-day history of centralized pleuritic chest pain which began when he was lifting boxes. The character is dull and constant. He also has back pain, most prominent when he wakes up, that has increased in severity over the past 3 days. Patient also stated he has had headaches that are localized to both his forehead and back of skull with no reported prodromal symptoms or known cause.
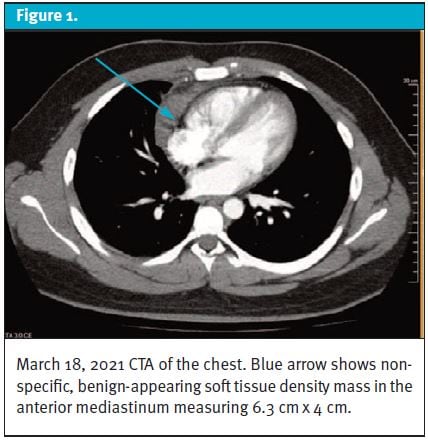
Patient was previously seen in the ED 3 weeks prior with multiple complaints, including right pleuritic chest pain that radiated to his neck and fever. A computed tomography angiography (CTA) of the chest (Figure 1), revealed a nonspecific, benign-appearing soft tissue density in the anterior mediastinum with differential of thymoma versus lymphoma. Patient was unable to seek recommended follow-up care.
Patient’s vitals revealed heart rate of 111 bpm and a temperature of 100.9°F. Patient’s labs were significant for a WBC: 15.5, glucose: 150, pro- calcitonin: 1.86, and an elevated D-dimer: 1.22. Three sets of troponins were drawn, all of which were negative. Patient’s COVID PCR was negative. EKG revealed sinus tachycardia. Chest x-ray showed prominence of the right heart border compatible with previously identified anterior mediastinal lesion. Thymic lesion vs lymphoma were considered.
Differential Diagnosis
GERD is unlikely, given the nature and duration of pain, and musculoskeletal pain was unlikely due to the lack of reproducible pain and location. Patient’s lack of history of connective tissue disorders, chronic hypertension, or severe, tearing chest pain made aortic dissection less likely. At patient’s age, lack of significant past medical history of hypertension/hyperlipidemia or common risk factors of immobility, recent surgeries, or trauma make ACS, PE less likely. Also, with a prior diagnosis of a mediastinal mass and the possibility of cancer-induced hypercoagulability, PE or ACS must be excluded. Although this patient was young and had no risk factors, prompt treatment is warranted for any patient with a high morbidity risk diagnosis, such as ACS, PE, or an aortic dissection.
CASE RESOLUTION
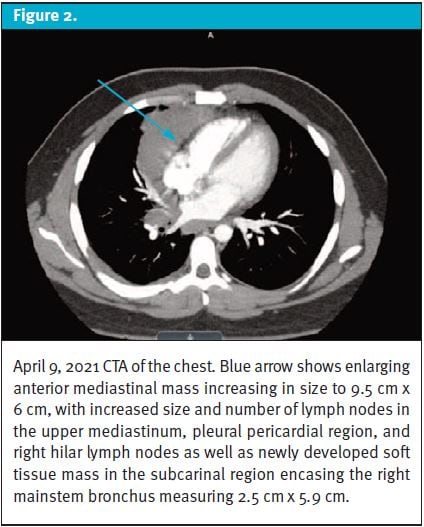
A repeat CTA in the ED 3 weeks later (Figure 2) showed a large mass measuring 9.5 cm x 6 cm in the anterior mediastinum that extended to the right of the midline, with an increase in size compared with 6.3 cm x 4 cm on previous CTA chest imaging (Figure 1).
A new soft tissue mass was visualized in the subcarinal region, with soft tissue encasing the right mainstem bronchus and newly enlarged right hilar and upper mediastinal lymphatic chain nodes and 1.4 cm enlarged pleuropericardial lymph nodes adjacent to the right ventricle, not visualized on previous imaging.
A CT scan of the abdomen and pelvis revealed heterogeneous pericardial and right perihilar infiltrate mass-like lesions with prominent pericardial and epicardial lymph nodes, the largest measuring 1.6 cm, as well as a 2.4 cm right paraesophageal lymph node lesion, most concerning for lymphoma.
Tissue samples for pathologic analysis were obtained on April 11, 2021 via endobronchial ultrasound bronchoscopy and on April 12,2021 via a CT-guided biopsy. Pathologic analysis revealed a diagnosis of T-cell lymphoblastic lymphoma. With diagnosis, patient was referred for prompt chemotherapy management.
DISCUSSION
As of 2018, 5,960 new cases and 1,470 deaths have occurred because of T-LBL in the United States.5 T-cell lymphoblastic lymphoma is a mediastinal mass that typically presents with shortness of breath due to compression of the superior vena cava.5
Although this disease more commonly presents in males and adolescents, further studies are needed evaluate whether there is a significant role for T-cell lymphoblastic lymphoma in people greater than 40 years of age.
In regard to this patient, there was a 6.3 cm x 4 cm mass that increased in size to 9.5 cm x 6 cm overtime. The increase in size was due to the presence of more than one extranodal localizations, with particular appreciation of lesions in the right hilar lymph nodes and upper mediastinal lymph nodes.6 Symptoms associated with back pain, as seen in this patient, can occur due to compression of nervous tissue structures from enlarged lymph nodes.

CONCLUSION
Although incredibly rare, T-cell lymphoblastic lymphomas can occur as a small subset of non-Hodgkin’s lymphoma. In this case report, we presented a patient with chest pain from a T-cell lymphoblastic lymphoma with a mass that had grown to 9.5 cm x 6 cm, compared with 6.3 cm x 4 cm on previous measurement (Figures 1 and 2).
This case may be utilized by medical professionals to draw attention to the importance of evaluating chest pain beyond the more common differential diagnoses or those that require prompt treatment. Evaluating younger patients further requires evaluation of other less common causes.
Manuscript submitted July 5, 2022; accepted September 9, 2022.
REFERENCES
- Ryu IH, Cho IS, Ryu AJ, et al. Long-term survival after T-cell lymphoblastic lymphoma treated with one cycle of hyper-CVAD regimen. Cancer Res Treat. 2015;47(1):115-119.
- Lepretre S, Graux C, Touzart A, et al. Adult T-type lymphoblastic lymphoma: treatment advances and prognostic indicators. Exp Hematol. 2017 Jul;51:7-16.
- Bassan R, Maino E, Cortelazzo S. Lymphoblastic lymphoma: an updated review on biology, diagnosis, and treatment. Eur J Haematol. 2016;96(5):447-460.
- Sweetenham JW. Treatment of lymphoblastic lymphoma in adults. Oncology (Williston Park). 2009;23(12):1015-1020.
- Hoelzer D, Gӧkbuget N, Digel W, et al. Outcome of adult patients with T-lymphoblastic lymphoma treated according to protocols for acute lymphoblastic leukemia. Blood. 2002;99(12):4379-4385.
- Coleman CN, Picozzi VJ Jr, Cox RS, et al. Treatment of lymphoblastic lymphoma in adults. J Clin Oncol. 1986;4(11):1628–37.
Author affiliations: Tushar Menon, MD, Abrazo Community Health Network. Sahil Kapur, DO, Arizona College of Osteopathic Medicine, Midwestern University. Ameera C. Mistry, MD, Arizona Medical Association. David M. Boyd, MD, Western Regional Medical Center. The authors have no relevant financial relationships with any commercial interests

