Published on
Urgent message: Needlestick injuries are common and require a prompt response based on an understanding of the latest USPHS guidelines and informed consent and counseling of the patient.
MAYA HECK, MS-2 and JOHN SHUFELDT, MD, JD, MBA, FACEP
Your center is fortunate to contract with a variety of different business to provide employee health services. A local homeless shelter is one of the clients to whom your center provides new hire physicals, drug screens and on the job injury care. A bright-eyed young volunteer arrives at your center from the shelter. While emptying the trash, she believes she poked herself with an exposed hypodermic needle. The hollow-bore needle went “all the way to my bone” before she pulled it out and doused her hand with rubbing alcohol. She is getting married in a month and wants to ensure she is “safe.” How do you respond?
Needlestick accidents and exposure to bodily fluids in health care and civil service settings are more com- mon than we’d like to think. As post-Affordable Care Act patient volumes increase, expect the number of needlesticks to increase as well as providers are more har- ried in their patient interactions. At the present time, according to the Centers for Disease Control and Pre- vention (CDC), about 385,000 sharps-related injuries occur annually among health care workers (health care workers) in hospitals.1 These data suggest that nearly 1 of every 10 health care workers in the United States has a needlestick exposure each year.2 It is speculated that even more go undocumented.
More than 30 different pathogens are known to cause infection following exposure to blood or body fluids in health care workers or hospital personnel.3,4 The most important of these pathogens, which are considered to be transmitted through a blood or secretion exposure, are hepatitis B virus (HBV), hepatitis C virus (HCV), and HIV infection.
In Part 1 of this two-part series, we’ll review the cur- rent CDC guidelines, the pathogenicity of the viruses, the pre and post exposure management of exposed health care workers, and preexposure prophylaxis and post-exposure management for hepatitis B and C. Part 2, in a subsequent issue, will review definitions of HIV transmission risk, HIV post-exposure prophylaxis, and the appropriate steps to follow for managing needle stick injuries.
Overview of CDC Recommendations
The CDC defines exposure as contact with blood, tissue, or other body fluids that may place a health care worker at risk of HIV infection and therefore requires consider- ation of post-exposure prophylaxis (PEP) as4:
- A percutaneous injury (eg, a needlestick or cut)
- Contact of mucous membrane or nonintact skin (eg, exposed skin that is abraded, or afflicted with dermatitis)
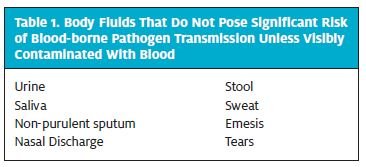
Body fluids of concern include: semen, vaginal secre- tions, or other body fluids contaminated with visible blood that have been implicated in the transmission of HIV infection, and cerebrospinal, synovial, pleural, peri- toneal, pericardial, and amniotic fluids, which have an undetermined risk for transmitting HIV. Table 1 lists body fluids that do not pose a significant risk of blood- borne pathogen transmission unless visibly contami- nated by blood.
Risk of infection is higher with exposure to: (1) a larger quantity of blood or other infectious fluid; (2) pro- longed or extensive exposure of non-intact skin or mucous membrane to blood or other infectious fluid or concentrated virus in a laboratory setting; (3) exposure to the blood of a patient in an advanced disease stage or with a high viral load; (4) a deep percutaneous injury; or (5) an injury with a hollow-bore, blood-filled needle. The CDC guidelines also mandate prompt evaluation for all potential exposures to blood or body fluids as defined previously. The name of the source, time/date of exposure, nature of exposure, body location and contact time with fluid, infective status of the source, and the description of injury should be obtained. In addition, obtaining a detailed history including dates of hepatitis B immunizations; previous testing for HIV, HBV, and HCV; tetanus immunization status; current medications; and current underlying medical condi tions should be recorded for the health care worker.
Provided that consent is given, all source cases should be tested for HBsAg, HCV, and HIV, unless the source is known to be infectious because subsequent pre-exposure prophylaxis (PEP) is based upon the results of source tests. PEP is recommended when occupational exposures to HIV occur. PEP should be initiated as soon as possible after the exposure. The general guidelines described above should always precede the steps that are specific to each pathogen and described later in this article.
If the source patient is known, test the source patient for hepatitis B surface antigen (HBsAg) and HCV and HIV antibodies. HIV viral load assessments for routine screening of source patients are NOT recommended. If available at the site of exposure, use a rapid HIV-anti- body test on the source patient. In some institutions, results are available in under 30 minutes. Rapid HIV tests approved by the US Food and Drug Administration are listed in Table 2.
If the source patient is NOT infected with a blood-borne pathogen, baseline testing or further follow-up of the health care provider is not necessary. The ability to dete mine the status of the source patient is “center dependent.” If you are unable to determine the individual’s sta- tus and the exposure occurs in a high-risk environment, consider using the two-drug PEP regimen as described in Table 3. State regulations related to informed consent and confidentiality also should be followed.
If the source patient is UNKNOWN, evaluate the like- lihood of high-risk exposure. Consider the likelihood of blood-borne pathogen infection among patients in the exposure setting, that is, what is the community infec- tion rate? Inquire about the setting in which the expo- sure occurred. Does the practice in which the needlestick or exposure occurred treat a large number of HIV-, HBV- or HCV-infected or at-risk patients? The general guide- lines stated above should always precede the following information specific to each pathogen.
Hepatitis B Virus
HBV is the most infectious of the three bloodborne viruses reviewed here for several reasons. Not only has HBV been transmitted by percutaneous and mucosal exposures, but also by fomites such as multi-dose med- ication vials, jet gun injectors, and endoscopes.5 In addition, HBV can survive and remain infectious on countertops for up to 7 days.6 The virulence of HBV has been greatly minimized since the advent of the HepB vaccine and Occupational Safety and Health Administration’s (OSHA) requirement that all health care workers with reasonably anticipated exposure to blood be offered the vaccine. Studies suggest that vaccination has been very successful, with a 95% decline in incidence of hepatitis B infection among health care worker between 1983 and 1995.7
Pre-exposure Prophylaxis. The Advisory Committee on Immunization Practices (APIC) and the Hospital Infections Control Practices Advisory Committee recommend that all health care workers with potential exposure to blood or blood products receive immunizations to protect against HBV.8,9 The approved dosing schedules are as follows:
- Engerix-B (Smith-Kline) — 0 mL
- (20 mcg) at 0, 1, and 6 months or 0, 1,
- 2, 12 months; or,
- Recombivax-HB (Merck) — 0 mL (10 mcg) at 0, 1, 6 months.
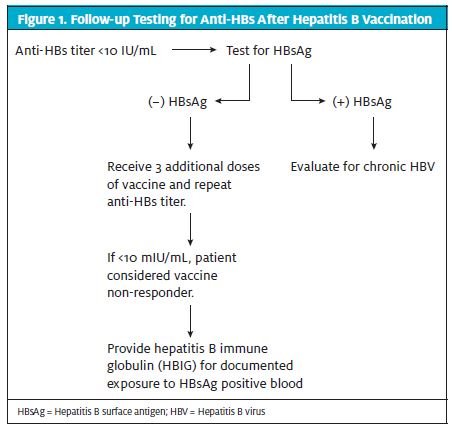
The Engerix schedule including 12 months is intend- ed for individuals who have or may have been exposed to HBV. In addition, follow-up testing for anti-HBs is required 1 to 2 months after the final vaccine dose, as illustrated in Figure 1.
Despite the decline in detectable vaccine-induced antibodies over time, booster doses are not recommended for immunocompetent health care workers.10 This is due to the protection provided against clinical hepatitis and chronic infection by the initial vaccine series even when their anti HBs levels become low or undetectable.
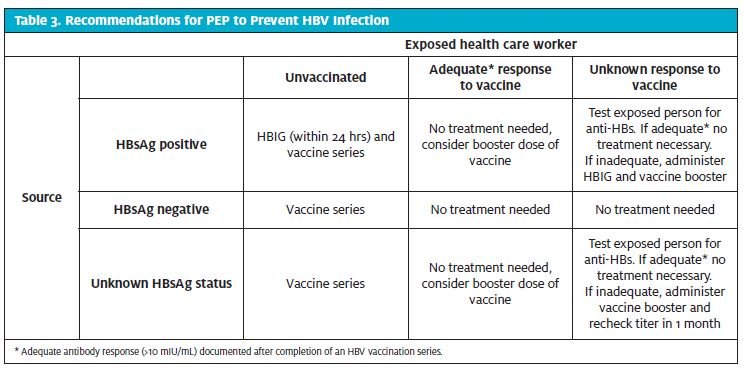
Post-exposure prophylaxis. PEP with HBIG and/or administration of the vaccine should be used after percutaneous or mucous membrane exposure to blood known or suspected to be HBsAg positive (Table 3).11
Hepatitis C Virus
Despite the awareness of transfusion-related HCV, the virus still remains a large health care burden. The asymptomatic nature of the disease increases the frequency of viral transmission among the population. Although the spread through transfusion products has been a leading cause of transmission of HCV, due to improved screening, it is now more strongly associated with intravenous and percutaneous drug and needle use.12
However, HCV is not transmitted easily through exposure of health care workers to blood; the average incidence of anti-HCV seroconversion after percutaneous exposure from an HCV-positive source is 1.8% (range: 0% – 7%).12 Pre-exposure Prophylaxis. Currently pre-exposure prophylaxis (PrEP) for HCV is not available. Post-exposure Prophylaxis. The Advisory Committee on Immunization Practices (ACIP) has concluded that the use of immune globulin (IG) as post-exposure prophylaxis after exposure (PEPE) to prevent HCV was not supported.13 Also, PEP use of interferon has not been demonstrated to reduce the rate of infection and in addition, interferon is associated with many side effects.14,15 The CDC recommends that individuals exposed to an HCV-positive source have the following baseline and follow-up testing16:
- Baseline testing for anti-HCV, HCV RNA, and alanine aminotransferase (ALT)
- Follow-up testing for HCV RNA 4 to 6 weeks after exposure
- Follow-up testing for anti-HCV, HCV RNA, and ALT 4 to 6 months after exposure
The National Institutes of Health (NIH) rec- ommends that exposed health care workers have a baseline immunoblot or HCV RNA assay.17 Aside from avoiding donating blood, plasma, organs, tissue, or semen during the follow-up period, individuals exposed to HCV-infected blood do not need to take any special precautions to prevent secondary transmission.18,19
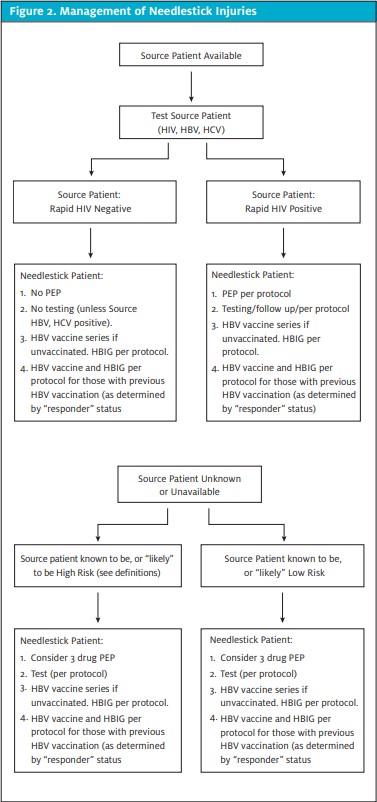
The algorithm in Figure 2 provides an overview of management of needlestick injuries. The National Clinicians’ Post-Expo- sure Prophylaxis Hotline is a 24/7 resource that offers advice on treatment and follow-up options. Call 1-888-448-4911 or visit http://www.ucsf.edu/hivcntr/PEPline.
The sidebar lists key points to remember when responding to a needlestick injury in a health care worker, so as to minimize the risk of legal liability.
Key Points in Response to a Needlestick in a Health Care Worker
- Timely evaluation and treatment of the exposed worker is
- Sources can only be tested after they have given informed consent.
- Providing accurate, written informed consent infor- mation to the patient is important and, if done appro- priately, will prevent the claim of “loss of a chance” if the patient sero-converts.
Conclusion
Although the treatment of patients who were exposed to a needlestick is nuance-driven, it is fairly straigh tforward if you simply follow the evidenced-base guide- lines. Every time a patient who has been exposed presents to an urgent care clinic, pull out the guidelines, get informed consent from the patient, provide written material, and treat the patient. In Part 2 of this article next month, we will discuss post-exposure guidelines for evaluation and treatment of HIV.
Click Here to download the article PDF.
References
- Centers for Disease Control and Prevention (CDC). The Stop Sticks Avail- able at:[http://www.cdc.gov/niosh/stopsticks/sharpsinjuries.html. Accessed August, 2013].
- Panlilio AL, Orelien JG, Srivastava PU, Jagger J, Cohn RD, Cardo NaSH Surveillance Group; EPINet Data Sharing Network. Estimate of the annual number of percutaneous injuries among hospital-based healthcare workers in the United States, 1997-1998. Infect Control Hosp Epidemiol. 2004;25(7):556-562.
- Katz MH, Gerberding Postexposure treatment of people exposed to the human immunodeficiency virus through sexual contact or injection-drug use. N Engl J Med. 1997;336:1097.
- Katz MH, Gerberding The care of persons with recent sexual exposure to HIV. Ann Intern Med. 1998;128:306.
- Henderson Management of needlestick injuries: A House Officer who has a needlestick. JAMA. 2012;307(1):75-84.
- Armstrong K, Gorden R, Santorella Occupational exposure of health care workers (health care workers) to human immunodeficiency virus (HIV): stress reactions and coun- seling interventions. Soc Work Health Care. 1995;21(3):61-8
- Kahn JO, Martin JN, Roland ME, et Feasibility of postexposure prophylaxis (PEP) against human immunodeficiency virus infection after sexual or injection drug use expo- sure: the San Francisco PEP Study. J Infect Dis. 2001;183(5):707-714.
- Gallant JE, DeJesus E, Arribas JR, et Tenofovir DF, emtricitabine, and efavirenz vs. zidovudine, lamivudine, and efavirenz for HIV. N Engl J Med. 2006; 354:251.
- Cohen HIV postexposure prophylaxis after sexual assault: why is it so hard to accomplish? Sex Transm Dis. 2008;35:979.
- Bassett IV, Freedberg KA, Walensky Two drugs or three? Balancing efficacy, tox- icity, and resistance in postexposure prophylaxis for occupational exposure to HIV. Clin Infect Dis. 2004;39:395.
- Seeff LB, Wright EC, Zimmerman HJ, et Type B hepatitis after needle-stick expo- sure: prevention with hepatitis B immune globulin: final report of the Veterans Admin- istration Cooperative Study. Ann Intern Med. 1978;88(3):285-293.
- Werner BG, Grady Accidental hepatitis-B-surface-antigen-positive inoculations: use of e antigen to estimate infectivity. Ann Intern Med. 1982;97(3):367-369.
- Bamberger JD, Waldo CR, Gerberding JL, Katz Postexposure prophylaxis for human immunodeficiency virus (HIV) infection following sexual assault. Am J Med. 1999;106:323.
- Cohen MS, Gay C, Kashuba AD, et Narrative review: antiretroviral therapy to pre- vent the sexual transmission of HIV-1. Ann Intern Med. 2007;146:591.
- Dumond JB, Yeh RF, Patterson KB, et Antiretroviral drug exposure in the female genital tract: implications for oral pre- and post-exposure prophylaxis. AIDS. 2007;21:1899.
- Van Damme P, Van Herck A review of the long-term protection after hepatitis A and B vaccination. Travel Med Infect Dis. 2007;5(2):79-84.
- S. Public Health Service. Updated U.S. Public Health Service Guidelines for the Man- agement of Occupational Exposures to HBV, HCV, and HIV and Recommendations for Postexposure Prophylaxis. MMWR Recomm Rep. 2001;50(RR-11):1-52. Available at: [http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5011a1.htm. Accessed August, 2013]
- Sy T, Jamal, M. Epidemiology of Hepatitis C Virus (HCV) infection. Int J Med Sci. 2006;3(2):41–46.
- Krawczynski K, Alter MJ, Tankersley DL, et Effect of immune globulin on the pre- vention of experimental hepatitis C virus infection. J Infect Dis. 1996;173:822—828.