Published on
Urgent message: The possibility of ear infection is the most common reason parents seek care for their young children during viral upper respiratory infections with fever. Urgent care providers should know the new criteria for making the clinical diagnosis and how to use technology such as an acoustic otoscope before concluding that antibiotics are necessary.
MICHAEL E. PICHICHERO, MD
Diagnosing acute otitis media (AOM) is a visual process based on viewing the eardrum and determining whether it is bulging, whether it is retracted or neutral in position, and whether there is a middle ear effusion.1 The examination is difficult to perform, and training in diagnosis often is limited and outdated. This review presents the newest guidelines on diagnostic criteria and treatment recommendations for AOM.
Epidemiology
AOM is an infection predominantly occurring among children between 6 months and 3 years of age.1–4 In nearly all cases, AOM is preceded by a viral upper respiratory infection (URI). AOM typically occurs on day 2 to 5 after onset of URI symptoms.1 Some viruses are more likely to lead to AOM, including respiratory syncytial virus, influenza, and rhinoviruses. Summer entero viruses very infrequently lead to AOM. Given these facts, the diagnosis of AOM without URI is so infrequent that a second look at the eardrum and reconsideration of the AOM diagnosis should be routine.1
Diagnosis
Medical History
The history of AOM is nonspecific and is essentially the same history as that of a viral URI (Table 1). The child has a runny nose, cough, and perhaps a fever. There may be crankiness, poor sleeping, and even tugging of the ear pinnae. None of the symptoms elicited in the medical history are diagnostic for AOM, because they all occur just as often as they occur during a viral URI. Therefore, most experts do not regard the medical history as particularly helpful incorrectly diagnosing AOM.1–4
Physical Examination
The Key Diagnostic Feature: A Bulging or Full Eardrum
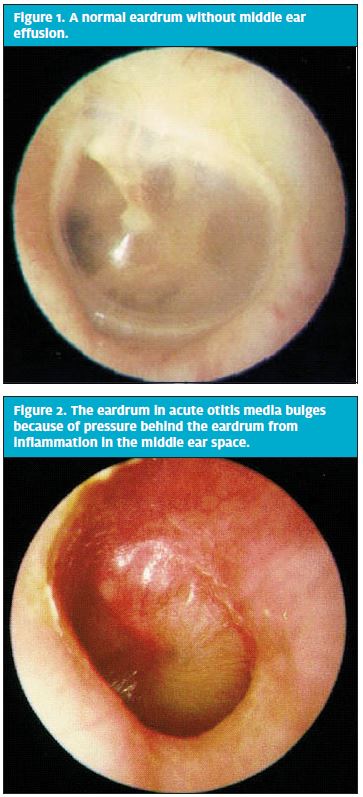
A normal eardrum without middle ear effusion behind it is shown in Figure 1. AOM should be diagnosed when the eardrum is bulging or full because of pressure behind the eardrum, caused by inflammation in the middle ear space, and when there is pus in the middle ear1 (Figure 2). This is a change from the past, when the presence of fluid behind the eardrum and redness of the eardrum were considered the important examination findings.
The switch to bulging as a key parameter for the diagnosis of AOM by the 2013 guidelines of the American Academy of Pediatrics (AAP) is evidence-based.2 Table 1 compares the sensitivity of various ear examination findings and the presence of bacteria behind the eardrum as proven by tympanocentesis.3 It is evident that bulging is the best predictor of bacteria causing AOM.
Redness of the eardrum is no longer considered a reliable sign because it has a poor correlation with bacterial AOM. Most cases of AOM occur in children between ages 6 months and 3 years of age, and they cry when their ears are examined. Crying, insertion of a speculum, and fever makes the eardrum turn red, so that is why redness has been removed as a key diagnostic sign of AOM. An exception is if the clinician sees a child whose one eardrum is bright red but whose other eardrum is of normal color.
Acute Otitis Media Is Not Associated with a Retracted Eardrum
A retracted eardrum is usually a viral-mediated process and is associated with otitis media with effusion1 (OME;
Figure 3).
In older children and adults, obstruction of the Eustachian tube may occur because of an upper airway allergy, but in young children a retracted tympanic membrane is most often due to a viral URI. What happens in the middle ear space is much the same as what happens in the nose during a viral URI. Congestion and mucus build up in the Eustachian tube, and this causes the natural air flow into the middle ear from the back of the throat to diminish—just like a stuffy or blocked nose. With the Eustachian tube closed, the air in the middle ear seeps across the eardrum, creating a small vacuum in the middle ear. Consequently, the eardrum becomes retracted, so a retracted eardrum is a normal finding in the context of a viral URI and does not indicate AOM. The problem is that a retracted and bulging eardrum can be difficult to distinguish from the normal state. In both cases, the light reflex is splayed and the eardrum looks distorted.
Otitis Media with Effusion
It was once thought that AOM could be distinguished from OME because OME was not associated with inflammation in the middle ear space. OME was instead considered to be caused by a buildup of fluid caused by natural mucous secretions in the middle ear. New research findings show there are inflammatory mediators in the middle ear fluid when OME is present. OME causes hearing loss, and there can be other symptoms. In fact, fluid in the middle ear is uncomfortable, and a patient may feel popping noises as air gains entry via the Eustachian tube into the middle ear space. This may cause the child to pull and tug at the ear or even cry. Pressure changes in the middle ear can occur during OME, and pressure changes that move the eardrum forward or backward cause excitation of nerve receptors in the eardrum to fire and cause pain. The crying of children in an airplane on takeoff and landing is caused by pressure changes in the cabin, not the sudden development of AOM or OME.
Middle Ear Fluid Occurs with Acute Otitis Media and Otitis Media with Effusion
When the Eustachian tube is congested or closed during a viral URI, mucus begins to build up in the middle ear space. In the middle ear, goblet cells are present that make mucus to keep the middle ear epithelial cells moist. If that normally present mucus does not have an escape path through the Eustachian tube, then it builds up and a middle ear effusion becomes visible. For this reason, it is not sufficient to use a middle ear effusion as a sole criterion for diagnosis of AOM. In about one-third of cases when a child between 6 months and 36 months of age has a viral URI, they will develop AOM. That means if a clinician is diagnosing AOM in more than two-thirds of children presenting with a viral URI and symptoms consistent with AOM, then they are almost certainly overdiagnosing.4 Also when an earache is reported, about one-third of the time it is OME and two thirds of the time it is AOM (Figure 4).
Is the Eardrum Opaque or Translucent?
A third aspect of the examination of the eardrum to consider is whether it is translucent or opaque. Note that in Figure 1 the eardrum is translucent, whereas in Figure 2 it is opaque; in Figure 3 it is again translucent. However, opacity occurs because inflammation of the eardrum is part of AOM. The eardrum is thickened because of the inflammation, making it difficult or impossible to determine whether there is fluid behind the eardrum. When an eardrum is bulging, it has an inflammatory middle ear effusion pushing it toward the examiner. A translucent or transparent eardrum almost never occurs with AOM.1–4 A retracted eardrum cannot represent AOM, but a retracted eardrum with fluid behind it represents OME.
Pneumatic Otoscopy
The 2013 AAP guidelines recommend the use of pneumatic otoscopy to distinguish a bulging eardrum from a retracted eardrum.2 Training in this technique is omitted in most residency curricula, but the skill can be mastered with practice by any clinician. Pneumatic otoscopy involves making a seal with the speculum and generating positive pressure on insufflation to see backward movement when an eardrum is bulging and negative pressure to see forward movement when an eardrum is retracted.
Obstruction of Eardrum View Because of Wax in the External Ear Canal
About half of children between the ages of 6 months and 36 months, when AOM is most common, have sufficient wax that removal is essential to get an adequate view of the eardrum. Clinicians should not be satisfied with a glimpse of a small portion of the eardrum, because that is not sufficient to make the diagnosis of AOM. It is recommended that the clinician take the time and make the effort to clear all or nearly all of the ear canal earwax to view all or nearly all of the eardrum. A soft plastic earwax-removal spoon is often effective.
Disposition and Follow-Up
The new AAP guidelines advocate consideration of appropriate antibiotics or watchful waiting in management of AOM.2 Follow-up is recommended if the tympanic membrane has ruptured, if there is a history of recurrent AOM, or if there is a history of hearing loss.
Technology to Aid Diagnosis
Newer Otoscopes
To expertly examine the eardrum and middle ear space, it is essential to have maximum light of an appropriate color focused on the eardrum. Newer otoscopes offer potential advantages compared with older otoscopes. A macro-view otoscope allows the clinician to view the entire eardrum without panning and to zoom in and focus the device’s magnification to accommodate their own vision (myopia and/or presbyopia). Also, newer otoscopes have improved the amount of illumination and color spectrum provided by the halogen lightbulb.
Tympanometers
A hand-held or desktop tympanometer unit may be used as an adjunct in examining the eardrum and middle ear.5–7 The tympanogram identifies the movement of the eardrum in response to positive and negative pressure, applied by pressing a button once a seal is established between the device speculum and the external auditory canal. If no movement occurs with applied pressure and the seal is adequate, then the tympanogram readout is flat (type B tympanogram). This is the typical case for AOM. In some cases the tympanic membrane will be full or bulging, also called a positive-pressure tympanogram readout (type A tympanogram). This occurs early in the pathogenesis of AOM. In some cases the tympanic membrane will be retracted, also called a negative-pressure tympanogram readout (type C tympanogram). This occurs during a viral URI, sometimes with concomitant middle ear fluid, and is consistent with OME. Tympanometry requires a seal of the inserted speculum-like device in the external auditory canal. In children younger than age 2 years, when AOM is most common, the child often moves during the use of the tympanometer and a seal cannot be obtained. Also, if a child is crying, a tympanogram reading cannot be obtained.
Spectral Gradient Acoustic Reflectometers
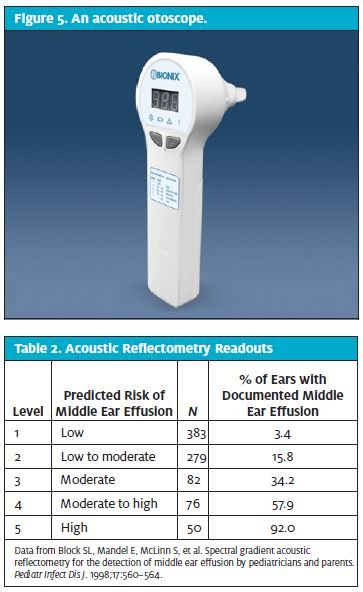
Another instrument helpful in the diagnosis of AOM and OME is the spectral gradient acoustic reflectometer (SGAR), also called an acoustic otoscope (Figure 5).
This technology assists the clinician in the detection of middle ear fluid.7,8 The device sends a sonar sound wave to detect middle ear fluid. If there is only air in the middle ear space, then the sonar sound wave bounces back, giving a high numeric readout on the head of the device. If the sonar sound wave bounces back slowly because of fluid in the middle ear, then the readout is a lower number. The more viscous the fluid in the middle ear, the slower the sonar sound wave bounces back. Thicker fluid is more likely pus associated with AOM, whereas thinner fluid is more likely associated with OME. The specificity of the SGAR to detect middle ear fluid7 is shown in Table 2.
The SGAR does not require a seal in the external auditory canal, so readings can be obtained in the crying child. The main limitation is the presence of wax in the external auditory canal. Complete blockage of the sonar wave emitted by the acoustic reflectometer by earwax produces a readout of “not interpretable,” whereas a blockage of 50% or less does not. SGAR use is not a replacement for otoscopy or pneumatic otoscopy. It is a diagnostic aid with a performance similar to that of tympanometry; the two technologies complement each other.
Red Flags for High-Risk Presentations
Any child with AOM who is younger than 1 month of age is at high risk, and the otopathogens affecting such a young child may be different. Consultation is advisable. AOM in the first 6 months of life is much more often associated with recurrent AOM than is AOM in the typical age range of 6 months to 36 months. Children with eardrum ruptures should be referred for follow-up examination. Fever is typical with URI and associated AOM, but high fever, in excess of 102°F (39°C), should prompt examination for other sources such as pneumonia.
Treatment
Changing Pathogens
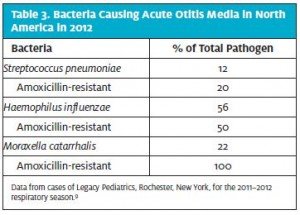
To prescribe the best antibiotic for AOM, it is important to be aware of the predominant otopathogens and their antibiotic susceptibility. The situation is dynamic because use of pneumococcal conjugate vaccines has caused a downward shift in prevalence of Streptococcus pneumonia and an upward shift in prevalence of Haemophilus influenza in recent years.9 Also, the proportion of S. pneumonia that are not susceptible to penicillin and the proportion of H. influenzae that are _-lactamase positive have been changing9 (Table 3).
Observation Option
The new AAP guidelines once again offer a noantibiotic treatment option.2 They still advocate antibiotics in the population of children who are very young and nonverbal.
Antibiotic Choices
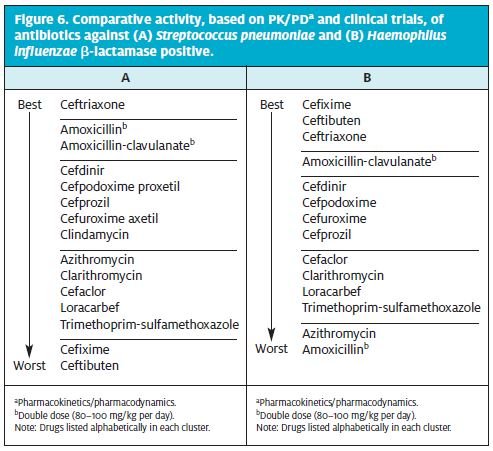
The 2013 AAP guidelines represent a change from the 2004 version regarding antibiotic recommendations.2 The continued recommendation of amoxicillin as the treatment of choice takes into account the frequency of overdiagnosis, the higher risk of serious infection by S. pneumoniae, and cost.2 Among the alternative treatments for AOM in children, in vitro testing allows a rank-ordering of their anticipated activity against the two most common otopathogens1 (Figure 6).
Symptomatic Treatment
Analgesics are recommended for pain and fever. Acetaminophen or ibuprofen in age-appropriate and weight-appropriate doses should be used. Use of analgesic eardrops is noted in the new AAP guidelines,2 but the evidence for their benefit is weak.
Management of Otitis Media with Effusion
OME resolves spontaneously within 3 months of onset in more than 90% of cases. Antibiotics, decongestants, antihistamines, and intranasal sprays of steroids or other products are not helpful.10
Disposition and Follow-Up
Guidelines from 2004 from the AAP, the American Academy of Family Physicians, and the American Academy of Otolaryngology—Head and Neck Surgery10 recommend that if OME does not resolve spontaneously within 3 months if involving both ears or within 6 months if involving one ear and there is associated hearing impairment, then referral to an otorhinolaryngologist for consideration of tympanostomy tubes is recommended.
Conclusion
Antibiotics should be prescribed for AOM but not for OME or otalgia. The key examination finding is the determination of the position of the eardrum and the presence of a middle ear effusion. To accomplish this task, the clinician must have adequate visualization of the eardrum and may supplement the examination with adjunctive tools such as pneumatic otoscopy, tympanometry, and acoustic otoscopy. Treatment may involve an observation option or an antibiotic. If an antibiotic is prescribed, then guidelines are available to direct selection. However, the situation is dynamic, so alternatives to traditional first-line choices should be assessed with each patient, taking into consideration the clinical condition and the efficacy, cost, and safety of the drug and the likelihood of good follow-up if the patient’s condition does not improve or worsens.
Conflict of Interest Statement
Dr. Pichichero has served as a consultant to Welch Allyn regarding development of newer otoscopes and tym panometers. He served part time from 2014 through 2015 as chief medical officer of a company that developed an improved SGAR; he no longer holds that position.
References
- Pichichero ME. Otitis media. Pediatr Clin North Am. 2013;60:391–407.
- Lieberthal AS, Carroll, AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131:e964–99. Available from: http://pediatrics.aappublications.org/content/131/3/e964.long
- Rothman R, Owens T, Simel DL. Does this child have acute otitis media? JAMA. 2003;290:1633–40.
- Pichichero ME. Acute otitis media: Part I. Improving diagnostic accuracy. Am Fam Physician. 2000;61:2051–6.
- Helenius KK, Laine MK, Tuhtinen PA, et al. Tympanometry in discrimination of otoscopic diagnoses in young ambulatory children. Pediatr Infect Dis J. 2012;31:1003–6.
- Chianese J, Hoberman A Paradise JL, et al. Spectral gradient acoustic reflectometry compared with tympanometry in diagnosing middle ear effusion in children aged 6 to 24 months. Arch Pediatr Adolesc Med. 2007;161:884–8.
- Block SL, Mandel E, McLinn S, et al. Spectral gradient acoustic reflectometry for the detection of middle ear effusion by pediatricians and parents. Pediatr Infect Dis J. 1998; 17:560–4.
- Laine MK, Tähtinen PA, Helenius KK, et al. Acoustic reflectometry in discrimination of otoscopic diagnoses in young ambulatory children. Pediatr Infect Dis J. 2012;31:1007–11.
- Casey JR, Kaur R, Friedel V, Pichichero ME. Acute otitis media otopathogens during 2008–2010 in Rochester, NY. Pediatr Infect Dis J. 2013;32:805–9.
- Rosenfeld RM, Culpepper L, Doyle KJ, et al; American Academy of Pediatrics Subcommittee on Otitis Media with Effusion; American Academy of Family Physicians; American Academy of Otolaryngology—Head and Neck Surgery. Clinical practice guideline: otitis media with effusion. Otolaryngol Head Neck Surg. 2004;130(5 Suppl):S95–118.