Published on
Urgent message: The prevalence of cancer is increasing—and along with it, malignancy-associated complications. Early recognition and management of these conditions is vital to alleviating patient morbidity and maximizing quality of life.
Introduction
Cancer is a leading cause of morbidity and mortality throughout the world, accounting for over 580,000 deaths in 2013 in the U.S.1 With an aging population and more effective forms of treatment, the overall prevalence of cancer is increasing. Consequently, acute cancer-related complications are more common.2 For many patients, an oncologic complication will be their initial manifestation of cancer.3 Urgent care providers will be increasingly exposed to complications of cancer and cancer treatments, and it is paramount to recognize and know how to manage patients presenting with acute cancer complications. Early recognition and management can alleviate morbidity and sustain quality of life. This review will provide an overview of the pathophysiology, manifestations, and management of five common acute malignancy-associated complications: febrile neutropenia, superior vena cava syndrome, malignant spinal cord compression, malignancy-associated hypercalcemia, and venous thromboembolism.
Case Presentation
A 73-year-old man presents to an urgent care center complaining of shortness of breath. He states that the shortness of breath has worsened over the past several weeks, along with a feeling of facial fullness and increased cough. He has a history of non‒small-cell lung cancer that is actively being treated with radiation and chemotherapy. Other than an oxygen saturation of 92% on room air, his vital signs are normal. The physical exam is notable for swelling of the face and elevated jugular venous distention.
This leads to the following questions:
- What diagnoses are important to consider in this patient who is undergoing chemotherapy and radiation therapy for an already-recognized cancer?
- How can these conditions be diagnosed?
- How should this patient be managed?
- When should patients presenting with suspected malignancy-associated complications be referred to a higher level of care?
Discussion
Febrile Neutropenia
Overview
Febrile neutropenia is a life-threatening complication of cytotoxic medications utilized to treat malignancy. The Infectious Diseases Society of America (IDSA) defines fever in neutropenic patients as a single oral temperature >38.3°C (101°F) or temperature >38°C (100.4 °F) for one hour.4,5 While cytotoxic medications destroy cancerous cells, they may cause bone marrow suppression. While all cell lines can be affected, the reduction in neutrophils is most clinically important. Most chemotherapy regimens result in a neutrophil nadir 7-10 days after treatment.6 Neutropenia is defined as an absolute neutrophil count (ANC) of <1500 cells/μL, with severe defined as an ANC <500 cells/μL, or an expected drop to <500 over 48 hours.5 As the severity and duration of neutropenia increases, the likelihood for the development of bacteremia increases, and with it, the progression to sepsis.7
In addition to bone marrow suppression, cytotoxic chemotherapy drugs may damage the mucosal lining of the gastrointestinal tract.4 This provides a portal for entry of bacteria and the development of bacteremia and septicemia.6 Approximately 80% of identified infections are thought to arise from the patient’s endogenous flora, with gram-positive sources most common.7 Gram-negative infections possess greater severity (specifically, Pseudomonas).7 Though less common, infections with fungal and viral pathogens also occur, more commonly after a prior episode of neutropenic fever.6
Patients with febrile neutropenia can be categorized into high and low risk groups. The Multinational Association for Supportive Care in Cancer (MASCC) risk index, (Table 1) is a validated tool utilized to calculate the risk of medical complications and to categorize patients.4 Scores above 21 place patients at low risk, while a score below 20 categorizes patients as high risk. Low-risk patients are those with mild or no symptoms, no hypotension or evidence of chronic obstructive pulmonary disease (COPD), solid tumors or hematologic malignancies, no previous fungal infection, and <65 years old.8 High-risk patients are those presenting with shock, ANC <500, ANC levels low for >7 days, or presence of organ dysfunction.8
| Table 1. MASCC Risk Index Score Breakdown | |
| Characteristic | Weight |
| Febrile neutropenia with no or mild symptoms | 5 |
| No hypotension | 5 |
| No COPD | 4 |
| Solid tumor or hematologic malignancy with no previous fungal infection | 4 |
| No dehydration requiring parenteral fluids | 3 |
| Febrile neutropenia with moderate symptoms | 3 |
| Outpatient status | 3 |
| Age <60 years | 2 |
(Adapted from http://www.mascc.org/mascc-fn-risk-index-score)
Presentation
All patients who have received chemotherapy within the last 4-6 weeks presenting with a fever or who generally feel unwell should be assessed for febrile neutropenia. Fever is often the earliest, and sometimes only, manifestation of an infection in these patients due to a diminished inflammatory response.6
Physical exam should focus on skin, catheter sites, oropharynx, sinuses, mucous membranes, heart, lungs, abdomen, and perianal area. A rectal exam should be avoided in these patients for fear of trauma to the rectal alimentary tract. The patient must be evaluated for any signs or symptoms of pneumonia. An abdominal exam should be performed to assess for any tenderness or peritoneal signs, which may signify typhlitis (necrotizing enterocolitis). Skin folds must be examined along with vascular access sites or surgical sites for erythema, tenderness, and discharge.4
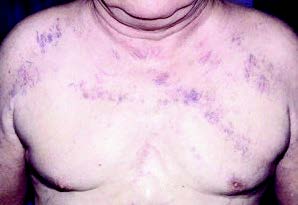
Figure 1. Collateral vessel formation with resulting ecchymosis in a
patient with SVC syndrome.(Source: http://www.emdocs.net/oncologic-emergencies-part-i-pearls-and-pitfalls/)
Management
While most patients undergoing chemotherapy have instructions from their oncologist to present immediately to the emergency department (ED) if they have fever or feel unwell, urgent care providers must be able to rapidly identify and manage these patients. First, the patient’s vitals (including temperature) should be obtained, along with intravenous (IV) access. Initial labs should include a complete blood count, blood cultures with lactate, urinalysis, renal function panel, and liver function panel.4 Additionally, an electrocardiogram (ECG) and chest x-ray should be ordered. Patients with pneumonia may or may not have a consolidation on chest x-ray due to a muted immune response.9
The most important part in the management of these patients is rapid treatment with antibiotics. It is recommended that antibiotics be initiated within 60 minutes of presentation.10 In the urgent care setting, any patient presenting with suspected neutropenic fever should be given fluids and started on antibiotics and transferred to the nearest ED with access to oncology. Table 2 shows antibiotic regimens for patients with neutropenic fever, based on risk assessment.
| Table 2. Antibiotic regimens for febrile neutropenia4,6-8,10 | |
| High risk | Broad-spectrum antipseudomonal penicillin plus aminoglycoside (hospital-dependent) |
| Low risk | Ciprofloxacin plus amoxicillin and clavulanate or Ciprofloxacin plus clindamycin |
| Skin/soft tissue/central line infection; pneumonia; mucositis; and/or shock present | Add vancomycin (if suspicious of gram-positive organism) |
Patients who identified as low risk may qualify to be treated on an outpatient basis. The success rate in these patients is around 80%, with 20% requiring readmission.4 Patients at increased risk include those older than 70 years, severe mucositis, poor performance status at home, and neutropenia <100 cells/μL.11 Collaboration with the patient’s oncologist is required in determining patient disposition.
Superior Vena Cava Syndrome
Overview
Superior vena cava (SVC) syndrome results from obstruction of blood flow through the SVC, which can be caused by either internal vascular invasion or external compression.12,13 The majority of cases are due to malignancy, but up to 40% are due to intravascular devices.14,15 The most common types of malignancy associated with SVC syndrome are non‒small-cell and small-cell lung cancer, followed by lymphoma.12,13
Presentation
Symptom onset and severity depend on the degree and rate of obstruction.13 Slower developing obstruction allows for venous collateral formation, which can decrease the severity of symptoms.6 The most common symptom is dyspnea; cough, chest and shoulder pain, hoarseness, and dysphagia may also occur.12,13 Signs of SVC syndrome include facial edema (most common), distended neck veins, superficial chest veins, arm edema, and facial plethora.15 While uncommon, SVC obstruction and/or neck edema can be severe enough to impinge the airway, leading to need for airway support.6,13 The urgent care physician must take note of this when examining the patient and carefully evaluate for signs of respiratory distress.
Figure 1. Collateral vessel formation with resulting ecchymosis in a patient with SVC syndrome. (Source: http://www.emdocs.net/oncologic-emergencies-part-i-pearls-and-pitfalls/)
Diagnosis
Chest x-ray is abnormal in 84% of patients with SVC syndrome, often showing widening of the mediastinum and pleural effusion. The optimal imaging study is a CT of the chest with contrast. Collateral vessel presence on CT has a specificity for SVC syndrome of 96% and sensitivity of 92%.13,15,16
Management
If SVC syndrome is suspected, the patient should be transferred to the ED. Initial management consists of sitting the patient upright, administering oxygen, and initiating steroids.6 Emergency management is usually not necessary, unless the patient has signs of airway compromise. The median survival period of patients with SVC syndrome due to cancer is about 6 months, but this is variable depending on the underlying malignancy.12,13,17
Malignant Spinal Cord Compression
Overview
Malignant spinal cord compression (MSCC) is a common cancer complication resulting from thecal sac impingement from an extradural mass.18 It is estimated to develop in about 5% of cancer patients.19 The three most common cancers leading to MSCC are lung, breast, and prostate cancer, each of which accounts for 20% of cases.20 Multiple myeloma, non-Hodgkin lymphoma, and renal cell carcinoma each account for 5% to 10% of cases.18 As the thoracic spine has the largest blood supply, the greatest number of vertebrae, and the least amount of space in the spinal canal, it is the most susceptible to compression.13 Approximately 60% of lesions occur in the thoracic spine, 30% in the lumbar spine, and 10% in the cervical spine.21
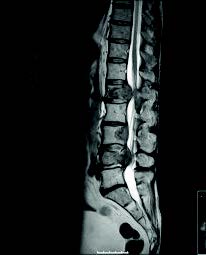
Figure 2. Vertebral metastasis leading to spinal cord impingement. (Source: www.medscape.com/viewarticle/442735)
Presentation
The most common symptom of MSCC is back pain, found in 80% to 95% of presentations; this often precedes the onset of other symptoms by several months.12,13 Pain is typically progressive and may be exacerbated by coughing, sneezing, or bending.6 Back pain that awakens a patient from sleep is concerning. While back pain is a common complaint in the urgent care setting, a presentation of back pain in a patient with a known cancer warrants investigation.
In addition to back pain, many patients will have neurological symptoms and signs. Weakness is present in up to 85% of patients and depends on the level of impingement. Sensory findings are less common than motor findings and may manifest as ascending numbness and/or paresthesias. Additionally, half of patients will have bowel and/or bladder dysfunction at presentation, but this is generally a later finding.13,22
Diagnosis
Any patient in whom MSCC is suspected must undergo urgent magnetic resonance imaging of the whole spine. Up to one third of patients will have multiple sites of metastasis and/or compression. In addition, post-void residual or ultrasound can be helpful during initial evaluation if bladder or bowel symptoms are present.13
Figure 3. MRI demonstrating multiple lesions in the lumbar spine with spinal cord compression. (Source: www.emdocs.net/oncologic-emergencies-part-i-pearls-and-pitfalls/)
Management
Prompt treatment of MSCC is key, as it can palliate pain and help prevent the progression of neurologic symptoms. If MSCC is suspected, the patient should be immediately transferred to a facility with a spine surgeon and oncologist. Before transfer, initial management in these patients consists of pain management and high-dose steroids if severe neurologic deficits such as paraparesis or paraplegia are present.13 Even with proper management, the prognosis for patients with MSCC is poor, with a median survival of 3-6 months and a 1-year survival rate of 30%.6
Malignancy-Associated Hypercalcemia
Overview
Malignancy-associated hypercalcemia (MAH) occurs in 20% to 30% of cancers.12,23 Calcium homeostasis is maintained by multiple mechanisms: intestinal absorption, bone resorption, and renal excretion. Parathyroid hormone (PTH) acts to increase calcium resorption from bone, increase activation of calcitriol (active vitamin D) from calcidiol (inactive vitamin D), and promote calcium absorption and phosphate excretion from the kidneys. The most common mechanism leading to MAH is secretion of parathyroid hormone-related protein (PTHrP) by tumor cells.6,12,13,23 PTHrP can be produced by squamous cell carcinoma and lymphoma and accounts for about 80% of MAH.13,23 The second most common mechanism of MAH is osteolysis resulting from bone metastases.6,13 The most common cancers with bone involvement are breast, lung, and multiple myeloma.13
Presentation
Symptoms are non-specific and include dehydration, polydipsia, fatigue, confusion, nausea/vomiting, constipation, and muscle weakness.13 The classic description of the symptoms from hypercalcemia is “stones, bones, groans, and psychiatric overtones.” Patients with severe hypercalcemia can present with life-threatening complications such as acute pancreatitis, acute renal failure, or coma.6 ECG changes can also occur, including bradycardia, prolonged PR, widened QRS, and/or shortened QT interval.24
Diagnosis
MAH can be categorized on the level of severity of the hypercalcemia, specifically the total serum calcium level: mild (10.5-11.9 mg/dL), moderate (12.0-13.9 mg/dL), or severe (≥14.0 mg/dL).12 In addition to calcium levels, other laboratory studies to order in these patients include PTH concentration, a complete blood count with differential, electrolytes, renal and liver function tests, and phosphate and magnesium.6,12 A chest x-ray is helpful, as squamous cell carcinoma of the lung is the most common cause of MAH.6
Management
Hydration is the cornerstone of treatment for hypercalcemia. Treatment of patients with hypercalcemia depends on severity. Patients with mild hypercalcemia with no symptoms can be sent home with instructions to hydrate and follow up with their oncologist. While patients with moderate hypercalcemia may not require immediate treatment, collaboration with the patient’s oncologist to determine course of action is warranted. Patients with severe hypercalcemia should receive initial treatment and be transferred to a higher level of care. Patients with symptomatic moderate to severe hypercalcemia should be initiated on crystalloids at a rate of 200-300 mL/hr.13 For patients with severe hypercalcemia, further treatment consists of calcitonin and bisphosphonates. Calcitonin is the fastest acting medication and is given at a dose of 4 IU/kg intramuscularly.13 The mainstay of therapy (besides hydration) is the bisphosphonates, pamidronate and zoledronate. These medications bind to hydroxyapatite and inhibit bone crystal dissolution and osteoclast resorption.25 In addition to calcitonin and bisphosphonates, patients may need dialysis if they have neurological deficits and a calcium level of ≥18 mg/dL.13 Loop diuretics are not indicated in these patients unless they have renal or heart failure.13 Unfortunately, the prognosis is poor in these patients, with half of all patients dying within a month of diagnosis of MAH.26
Venous thromboembolism
Overview
The pathogenesis of venous thromboembolism is outlined by Virchow’s triad, consisting of alternations of blood flow (stasis), vascular endothelial injury, and a hypercoagulable state. Patients with cancer are at an increased risk of venous thromboembolism (VTE) due to a hypercoagulable state stemming from production of procoagulants.27 VTE is estimated to be clinically significant in up to 15% of cancer patients.28 Risk factors are detailed in Table 3.
| Table 3. Risk Factors for VTE4 | |
| Patient-related | Age, obesity, history of smoking, decreased mobility |
| Cancer-related | Type of cancer*, stage, histologic type, |
| Treatment-related | Anticancer medications, radiation, history of recent surgery, presence of peripheral line |
| Biochemical-related | Hemoglobin <10, WBC>11×109/L, platelets >350×109/L |
*Brain, stomach, lung, pancreas, renal, uterus, and bladder cancers have highest rate of VTE.
The Khorana score, shown in Table 4, is well-validated in the cancer-patient population. Literature supporting the use of Wells or Geneva in this population is lacking.29
Table 4. Khorana Score
| Risk Factor | Points |
| Primary tumor site | 2
1 0 |
| Very high risk: stomach, pancreas | |
| High risk: lung, lymphoma, gynecologic, bladder, testicular | |
| All other sites | |
| Prechemotherapy platelets count ≥350,000/μL | 1 |
| Hemoglobin level <10 g/dL or use of RBC growth factors | 1 |
| Prechemotherapy WBC >11,000/μL | 1 |
| BMI ≥ 35 kg/m2 | 1 |
(Adapted from http://www.current-oncology.com/index.php/oncology/article/view/1938/1537, http://www.uptodate.com.proxy.library.vanderbilt.edu/contents/image?imageKey=HEME%2F73002&topicKey=HEME%2F1352&rank=1~2&source=see_link&search=khorana+score&utdPopup=true)
Presentation
There are two major clinical manifestations of venous thromboembolism in the cancer population: deep vein thrombosis (DVT) and pulmonary embolism (PE). A DVT occurs when a thrombus forms in one or more deep veins. PE occurs when the thrombus embolizes to the pulmonary arteries and occludes blood flow from the right ventricle. DVTs can be difficult to diagnose, as many of the classic signs and symptoms may not be present. The classic presentation of DVT is swelling, pain, and erythema of the affected leg.30 These are suggestive for DVT but not diagnostic. Homan’s sign of pain on dorsiflexion of the foot with an extended knee is classic, but not reliable.30
Clinical suspicion is often based on the history and risk factors rather than the presentation. PE can present with a wide variety of symptoms. The classic presentation is a patient with dyspnea, tachypnea, and chest pain.4 Patients may be asymptomatic and, as in DVT, clinical suspicion should be based on the presence of risk factors.
Diagnosis
VTE suspicion warrants transfer to a higher level of care. This facility must possess the necessary equipment to evaluate for VTE and subsequently manage this patient in an inpatient setting. If patient’s presentation is concerning for PE, CT scan of the chest to assess for is necessary. A CT pulmonary angiogram is the definitive form of imaging to evaluate for a PE.31 For DVT, in most situations compression ultrasonography is the preferred method for diagnosis.4
Management
The mainstay of therapy for VTE is anticoagulation with low molecular weight heparin.4 Anticoagulation should be initiated immediately, as delay can lead to embolization.32 Most patients will require admission. Outpatient therapy is not appropriate in patients with massive DVT, suspected PE, high bleeding risk, or other comorbidities.33 It is essential to transfer these patients to an ED.
Case Resolution
The 73-year-old man whose case was described at the start of this article presented with signs and symptoms concerning for SVC syndrome. While dyspnea in this patient is also concerning for PE, several factors in the presentation, including the insidious onset of symptoms, are more suggestive of SVC syndrome. The patient was positioned upright and given supplemental oxygen and dexamethasone 4 mg IV. Chest x-ray demonstrated a widened mediastinum and pleural effusion. The urgent care provider called the patient’s oncologist and explained his suspicion of SVC syndrome and the need to rule out PE. They agreed to transfer the patient to a center where he could receive imaging, exclude a PE, and initiate the work-up of his probable SVC syndrome.
Conclusion
As the prevalence of cancer increases, urgent care clinicians can expect to see more patients presenting with complications. Recognition and early management of these are vital while expediting transfer to a higher level of care. Of particular note in the urgent care setting:
- Febrile neutropenia is a concern in any patient undergoing chemotherapy, and antibiotics must be initiated promptly.
- SVC syndrome is a common complication of several types of cancer, and rapid identification can maintain a higher quality of life in these patients.
- Malignant spinal cord compression must be ruled out in any patient with a previously diagnosed cancer who presents with back pain; diagnosis and management can prevent neurologic impairment and disability.
- Malignancy-associated hypercalcemia occurs in approximately one quarter of cancer patients; fluids and bisphosphonates are the mainstays of treatment.
- VTE is a potentially life-threatening complication of cancer, and recognition and evaluation can be lifesaving.
While most of these oncologic emergencies are associated with a poor prognosis, rapid recognition and management are vital to minimize morbidity and sustain quality of life for these patients.
Drew A. Long, BS, Brit Long, MD, and Alex Koyfman, MD
Citation: Long DA, Long B, Koyfman A. An urgent approach to malignancy complications. J Urgent Care Med. January 2017. Available at: https://www.jucm.com/urgent-care-approach-malignancy-complications/.
References
- Long B. Oncologic emergencies part II: pearls and pitfalls. emDocs. August 21, 2015. Available at: http://www.emdocs.net/oncologic-emergencies-part-ii-pearls-and-pitfalls/.
- Walji N, Chan AK, Peake DR. Common acute oncological emergencies: diagnosis, investigation and management. Postgrad Med J. 2008;84:418-427.
- Bow E, Wingard JR. Overview of neutropenic fever syndromes. UpToDate. Oct 2015.
- Helman A. Episode 33: Oncologic Emergencies. Emergency Medicine Cases. http://emergencymedicinecases.com/episode-33-oncologic-emergencies/.
- Sipsas NV, Bodey GP, Kontoyiannis DP. Perspectives for the management of febrile neutropenic patients with cancer in the 21st century. Cancer. 2005;103:1103-13.
- Flowers CR, et al. Antimicrobial prophylaxis and outpatient management of fever and neutropenia in adults treated for malignancy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31(6):794.
- Escalante CP, et al. Outcomes of treatment pathways in outpatient treatment of low risk febrile neutropenic cancer patients. Support Care Cancer. 2004;12(9):657-662.
- McCurdy MT, Shanholtz CB. Oncologic emergencies. Crit Care Med. 2012;40(7): 2212-2222.
- Long B. Oncologic emergencies part I: pearls and pitfalls. emDocs. 13 August 2015. http://www.emdocs.net/oncologic-emergencies-part-i-pearls-and-pitfalls/.
- Rice TW, Rodriguez RM, Light RW. The superior vena cava syndrome: clinical characteristics and evolving etiology. Medicine (Baltimore). 2006;85(1):37.
- Reed ED, Dmitry JR. Malignancy-related superior vena cava syndrome. UpToDate. Oct 2015.
- Parish JM, Marschke RF Jr, Dines DE, et al. Etiologic considerations in superior vena cava syndrome. Mayo Clin Proc. 1981;56(7):407.
- Yellin A, Rosen A, Reichert N, et al. Superior vena cava syndrome. The myth—the facts. Am Rev Respir Dis. 1990;141(5 Pt 1):1114.
- Schiff D. Spinal cord compression. Neurol Clin. 2003; 21:67-86; viii.
- Bach F, Larsen BH, Rohde K, et al. Metastatic spinal cord compression: occurrence, symptoms, clinical presentations and prognosis in 398 patients with spinal cord compression. Acta Neurochir (Wien). 1990;107:37-43.
- American Cancer Society: Cancer facts and figures. Atlanta, GA; 2010.
- Klimo Jr P, Thompson CJ, Kestle JRW, et al. A meta-analysis of surgery versus conventional radiotherapy for the treatment of metastatic epidural spinal disease. Neuro-oncol. 2005;7:64-76.
- Helweg-Larsen S, Sorensen PS. Symptoms and signs in metastatic spinal cord compression: a study of progression from first symptom until diagnosis in 153 patients. Eur J Cancer. 1994;30A(3):396.
- Stewart AF. Clinical practice: hypercalcemia associated with cancer. N Engl J Med. 2005;352(4):373.
- Diercks DB, Shumaik GM, Harrigan RA, et al. Electrocardiographic manifestations: electrolyte abnormalities. J Emerg Med. 2004;27(2):153.
- Fleisch H. Bisphosphonates: pharmacology and use in the treatment of tumour-induced hypercalcaemic and metastatic bone disease. Drugs. 1991;42:919-944.
- Ralston SH, Gallacher SJ, Patel U, et al. Cancer-associated hypercalcemia: morbidity and mortality. Clinical experience in 126 treated patients. Ann Intern Med. 1990;112:499-504.
- Bauer KA, Lip, GY. Overview of the causes of venous thrombosis. UpToDate. Oct 2015.
- Chew HK, Wun T, Harvey D, et al. Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch Intern Med. 2006;166(4):458-464.
- Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111(10):4902.
- Bauer KA. Approach to the diagnosis and therapy of lower extremity deep vein thrombosis. UpToDate. Oct 2015.
- Thompson BT, Hales CA. Overview of acute pulmonary embolism in adults. UpToDate. October 2015.
- den Exter PL, van Es J, Erkens PM, et al. Impact of delay in clinical presentation on the diagnostic management and prognosis of patients with suspected pulmonary embolism. Am J Respir Crit Care Med. 2013;187(12):1369-1373.
- Douketis JD. Treatment of deep vein thrombosis: which factors determine appropriate treatment? Can Fam Physician. 2005;51:217.
