Published on
Urgent message: The patients most prone to sexually transmitted diseases are also mostly likely not to have a primary doctor. Many will turn to urgent care for help.
THOMAS SUNSHINE, MD, FACOG
As urgent care physicians, we often are presented with patients who are worried that they may have a sexually transmitted disease or infection (STD/I). The number of yearly visits is difficult to estimate because the diagnosis codes of the visits vary. The Center for Disease Control’s (CDC) surveillance data estimates that there are 19 million new cases of STD/I yearly; many will go undetected or unreported.1 In 2009, the latest year for which statistics are available, there were about 1.5 million actual reported cases of chlamydia and gonorrhea. The 2006-2007 CDC data for the diagnosis group “unspecified viral and chlamydial infection” showed roughly 6 million ambulatory care visits. It is estimated that almost half of new STD/I cases will be in the 15-24-year-old age group, individuals who may not have an established primary care provider. As urgent care providers, we are therefore in the unique position to be the first to encounter many such patients, and a fundamental understanding of symptoms, testing processes, risk factors, treatments, and counseling are important. There is no formal categorization of what is included in a STD/I screening. This article focuses on specific infections that are important in urgent care.
Clinical Approach
The key components of the history include assessing the patient’s risk factors for STD/I exposure (Table 1). Do not assume heterosexual, homosexual, or bisexual contact. Simply ask. Explain that this is an universal approach and although occasionally awkward, it avoids having to make judgments about the patient’s sexual preferences.
A patient may not recognize prior symptoms as being indicative of the presence of an STD/I. An ulceration or sore area is often assumed by the patient to be an ingrown hair or associated with shaving. It is also common to hear a condyloma referred to as a skin tag. HPV infection may also be inferred from a female partner with a history of abnormal pap smears. Sore areas in the groin may represent adenopathy but may have been thought to be pulled muscles. Any unexplained systemic illness should also be investigated, with special attention paid to unexplained rashes and hepatic or genitourinary symptoms.

The standard general physical exam, including the genitalia, will usually suffice. Pay special attention to evidence of current rashes, jaundice, adenopathy, hepatic enlargement, abdominal tenderness, or skin ulcerations. In order to be prepared for specific testing during the genital exam, it is helpful to have the discussion ahead of time regarding which specific tests could be performed. Educating the patient about potential infections and their testing processes often takes some time. After this review, for financial reasons, the patient may only wish to test for certain infections. Cost-effective testing is important in the urgent care setting, as we are often faced with self-pay patients or those with high out-of- pocket coverage. Some tests take weeks to months to show evidence of infection, so follow up instruction should be documented.
An in-depth review of each infection and advanced treatments are beyond the scope of this article. The information provided here is intended to allow an informative discussion with patients and meet their need for STD screening, treatment choices, and counseling. The testing processes described are commonly used, but individual urgent cares should consult with their laboratories to learn what is recommended.
Human Papilloma Virus
There roughly 150 viral subtypes of human papilloma virus (HPV) causing human infection; about 40 subtypes are associated with the anogenital infection. Classification is based on their oncogenic potential. The low-risk subtypes cause condyloma (warts) and are not associated with the development of cancer. High-risk subtypes are associated with the development of cancer.
The natural history of this progression is complex and not fully understood. In fact, 80%-90% of infections may resolve on their own.2 No reporting process is formally established for HPV infections. The CDC estimates that about 20 million people are infected with a high-risk subtype of HPV (primarily type 16 or 18), with 6 million new cases a year. A National Cancer Institute study, pre-vaccine era, reported a prevalence of 27% in women ages 14-59 years old, with almost 45% in women ages 14-19.3 Surveillance data suggest an overall HPV prevalence of 23% of the high-risk subtypes and range from 35% in women aged 14-19 years of age to 6% in women 50-65 years of age.4 Such information emphasizes the widespread nature of HPV infection, with an estimated 357,000 annual office visits.5

The testing process for HPV involves collection of cervical cells, often at the time of a pap smear, which are analyzed for HPV DNA. Routine HPV testing is not generally recommended for screening but is performed reflexively if pap smear results justify it. If warranted, pap smear and HPV testing can be done as part of the STD/I exam. HPV infection can be latent, meaning there is cellular infection but viral amounts are so small that no skin changes have occurred and testing may not even detect their presence.6 Screening for HPV DNA is not indicated for males. In the absence of visible lesions, penile “colposcopy” (using 3%-5% acetic acid) can be performed, although this controversial. Since HPV can be latent or subclinical, a positive test result does not imply recent infection.
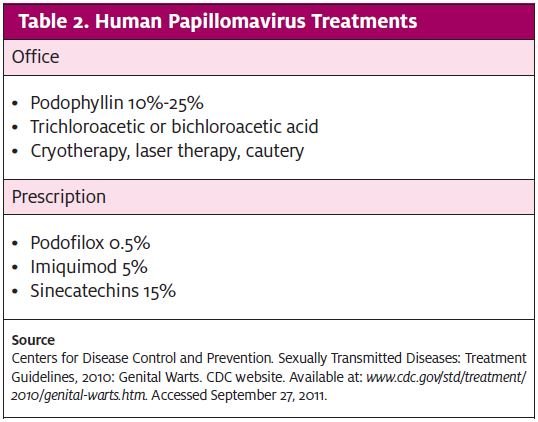
Treatment for HPV infections involves removal of visible condylomatous warts and an assessment of oncologic changes. Patients may receive prescription management or treatment in urgent care, depending on provider training (Table 2). More aggressive treatments involve excision, laser therapy, or interferon injections. Appropriate patients should be offered vaccination (Table 3).
Herpes
Herpes Simplex virus (HSV) type 1 is generally considered to cause outbreaks of the face and lip and type 2 of the genital region. But as a result of sexual practices, considerable overlap exists and either virus can be found at either site. Following initial infection, either virus establishes latency in either the trigeminal or sacral ganglia.
As with HPV, no specific reporting requirement exists for infection with HSV. However, it has been estimated that in 2009, there were about 300,000 initial outpatient visits for genital herpes.5 Based on antibody seropositivity, it is estimated that 50% of teens are positive for HSV 1, and the rate goes up to 85% by age 50 years. For HSV 2, in 14-49-year-olds, positivity is 16%. Similarly, 19% of people report a history of genital herpes based on survey reports rather than antibody titers. Contributing to such high infection rates are asymptomatic shedding rates of 5%-10% for both types 1 and 2.7
The high prevalence rates and often subclinical nature of the disease makes the decision to test for HSV in the urgent care setting a dilemma. Some patients will want to know if a certain sexual encounter infected them with HSV. In addition, many people do not realize there is no cure, just treatments to reduce the frequency of outbreaks or speed their healing. Data currently reflects that early treatment with an antiviral after initial exposure limits the viral amount eventually found in the ganglia, but it does not prevent disease. These are important issues to discuss with the patient presenting for STD testing who has no appreciable lesion. With a lesion present, the patient may be satisfied with a diagnosis by experienced visual inspection, but other STD/I’s can also cause ulcerations.
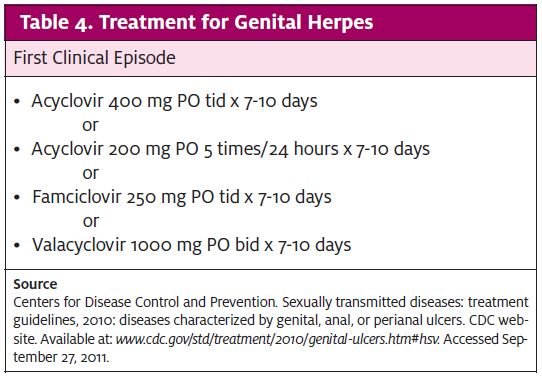
HSV immunoglobulin (Ig) M is positive at 7-10 days, and IgG becomes positive, depending on the test, as soon as 3-4 weeks. Because recurrent infection can elevate IgM as well, this nullifies IgM’s usefulness as an indicator of primary infection. A potentially predictive testing process would be an initial, negative, type-specific IgG that subsequently turns positive. It is important the patient has a clear understanding of the limitations of the testing process and results before proceeding. The clinician can do a viral culture on an open sore. The usefulness of a culture once a lesion is healing is limited. Also, a sample from the lesion may detect the presence of viral DNA using a process called “polymerase chain reaction.” CDC treatment guidelines are reviewed in Table 4.
It is important to consider screening for viral hepatitis in any STD/I visit. The important viral types in the United States are hepatitis A (HAV), B (HBV), and C (HCV). Viral types D and E are more prevalent in other parts of the world. Of these types, only HAV is passed via a fecal/oral route. The others are passed primarily by infected body fluids, which, in the context of STD/I screening, means semen and vaginal fluids, although percutaneous spread of infected blood is by far the most common route. The prevalence of these infections is recorded annually, based on voluntary reporting. It is thought that there is significant under-reporting.
Hepatitis A
In 2009, the CDC estimates there were 21,000 new cases of HAV infection. Considering the route of transmission, only certain sexual practices will pass this virus. Gastrointestinal and hepatic symptoms begin roughly 2-6 weeks following infection and are generally self-limited. Some people will have symptoms longer. HAV vaccination is recommended for individuals of all ages over 1 year old.
Following a specific exposure, the CDC recommended in 2007 that HAV vaccine be given to prevent infection, preferably within 2 weeks of exposure, in patients between 1 and 40 years of age. After age 40 years, based on limited vaccine data, immune globulins are the only intervention to prevent disease. Routine pre-vaccination screening for immunity is only recommended for groups likely to already have immunity; this is for cost control rather than medical issues. STD screening therefore represents a good time to offer HAV vaccination, and this should be included in any counseling.
Hepatitis B
The annual number of new infections of HBV is much greater than with HAV, with an estimated 38,000 annual new infections. Because HBV can develop into a chronic infection, roughly 800,000 to 1.4 million persons are thought to be chronically infected in the US. It is difficult to know how many of those cases are from sexual contact, as about 40% of people don’t know how or when they contracted the virus. Most individuals who have contracted the disease as an adult will recover and not become chronic carriers. Sexual transmission occurs from HBV virus present in semen or vaginal fluids or from percutaneous blood exposure; HBV is highly infectious. The higher an individual’s viral load, the higher the infectivity. The virus has been detected in saliva, tears, and urine, but these are not thought to be methods of transmission.
For an STD/I screening visit, the patient may present before the onset of gastrointestinal or hepatic symptoms, which begin an average of three months following exposure. The patient needs to know that he can be infectious weeks before developing any symptoms. If the patient has no specific history of vaccination, then vaccination should be initiated. (CDC treatment guidelines for hepatitis B are available at
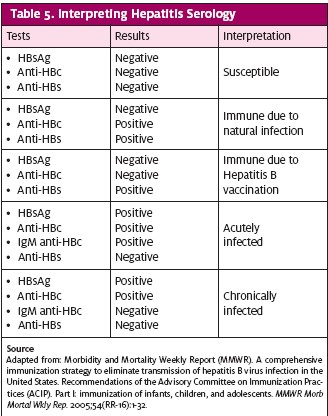
www.cdc.gov/std/treatment/.) Beginning vaccination within the first 24 hours after contact may prevent infection. There is no contraindication to giving the vaccine if the patient has already been vaccinated, assuming no previous allergic reaction. Universal vaccination at birth was first recommended by the Advisory Committee on Immunization Practices (ACIP) in 1991. Apart from counseling and vaccination, should the patient or provider need to know actual HBV status, blood serology can be performed. Table 5 provides a guide for interpretation of results.
Hepatitis C
HCV has the distinction of being the most common blood-borne infection in the US, with an estimated 3.2 million individuals currently infected. In 2009, however, only 16,000 people were thought to be newly infected; about 80% of them go on to become chronic carriers. HCV is substantially less infectious than HBV, making sexual transmission of HCV unlikely but not impossible. The CDC’s Morbidity and Mortality Weekly Report (MMWR) reported that 10% of newly infected people identified an infected sexual partner as the probable source.8 However, a study over 10 years of 895 monogamous couples, in which one partner had HCV, identified no partner-to-partner transmission.9 There is no vaccination or prophylactic treatment available and thus no specific interventions at the urgent care level. Counseling should focus on risk factors before deciding whether serologic testing for HCV antibody is warranted for the patient’s current status. Follow-up testing would depend on the development of hepatic (or other) symptoms in approximately three months.
Human Immunodeficiency Virus
Considering the vast amount of literature concerning HIV and its associated disease AIDS, it may be the patient’s primary concern. The most recent CDC data (2009) estimated that 48,000 people were diagnosed with a new HIV infection in the US. There were about 1.1 million people with HIV infection and 20% were unaware of their infection.10 Fifty percent of new infections were from male-to-male sexual contact and 30% were from heterosexual contact. This data does not translate well into probabilities of transmission from a single contact, as other factors—such as type of sexual practice, sex of the infected partner and the partner’s viral load, and presence of coexistent diseases—all impact the risk.11
Following an exposure to HIV, antibodies can develop as early as two weeks and in most patients by three months. Rapid, qualitative HIV antibody testing can be performed on blood or oral fluid in a Clinical Laboratory Improvement Amendments (CLIA)-waived laboratory in an urgent care. Any positive results from this immunoassay testing should be confirmed by the Western blot method before telling the patient of a confirmed diagnosis for HIV infection. If the patient tests negative, the provider should consider whether postexposure prophylaxis (PEP) should be initiated. PEP should be initiated within 72 hours after high-risk exposure, such as sex with a known carrier of HIV. With unknown partner status or presentation greater than 72 hours, there are no specific recommendations for PEP.
Gonorrhea
Neisseria gonorrhea (NG) is the second-most-commonly reported STD/I in the US, with 301,000 actual reports and 700,000 estimated infections occurring in 2009.
NG generally infects the genital tract, but be alert for infection of the rectum, eyes, oropharynx, skin, or joints. Following exposure, the chance of being infected from heterosexual sexual contact ranges from 20% if the infected partner is female to up to 70% if the infected partner is male.12,13 Patients can develop genitourinary symptoms as early as two to five days, although men may take up to one month. The patient is infectious all this time. Up to 80% of women and 10% of men may be asymptomatic.13
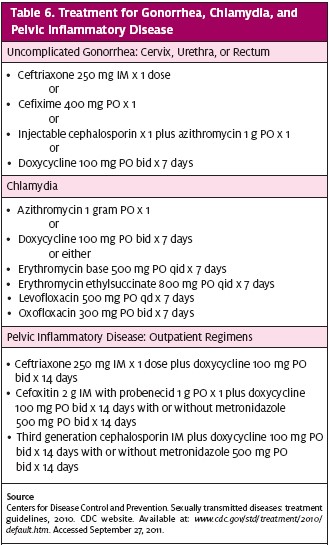
Testing for NG is conducted by swabbing the site of infection and performing either a nucleic acid amplification test (NAAT), culture, or gram stain. Many centers prefer to do NAAT on a urine specimen for genitourinary infection. (Urine testing for NG in women is not very sensitive, although it is a reasonable option for men.) The associated laboratory may not recommend NAAT on rectal, pharyngeal, or conjunctival swabs. When antibiotic sensitivity information is necessary, a culture should be performed. If a center provider has the appropriate ability, a gram stain from any infected sites to check for gram-negative diplococci can be performed, but this will have its primary diagnostic value for male urethral discharge.
Current treatment is specified by the CDC, as well as by regional antibiotic resistance data; the latter is available from local health departments. Due to the high rates of co-infection with chlamydia, an antibiotic regimen that treats both sexual partners is advocated. The most recent 2010 CDC recommendations for treatment are shown in Table 6. The CDC recommends that patients with a severe penicillin allergy have a consultation with an infectious disease specialist and may receive a single dose of 2 grams of azithromycin.
Chlamydia
Infection with chlamydia trachomatis is the most common bacterial STD/I reported to the CDC, with 1,244,000 infections in 2009, with an estimated number of over 2.8 million infections annually. The rates of chlamydia infection in 15-24-year olds ranges from 3.5% (Vermont) to 13.7% (Mississippi). This younger group has three times the rates of 25-49-year-olds. The most common symptoms are genitourinary, although chlamydia can infect the oropharynx, eyes, or rectum as well. Symptoms may occur as early as one week; however, up to two-thirds of women and one-half of men may not have symptoms.
A chlamydia test should be part of any STD/I screen whether a specific exposure can be identified or not. Universal screening is recommended for all sexually active females under age 25. NAAT can be performed on urine or with a cervical or urethral swab. NAAT testing methods are generally preferred in the urgent care setting and are done in conjunction with NG testing. Some laboratories can use liquid-based cytology specimens used for pap smears for NAAT testing as well.
Syphilis
Caused by the spirochete, Treponema pallidum, syphilis is part of any STD/I evaluation. Of 44,800 total cases in 2009, about 30,000 were diagnosed serologically, while in a latent stage. About two-thirds of the cases were reported from the male homosexual population. Symptoms, when occurring, appear primarily as painless chancres or secondarily as generalized symptoms affecting almost any body system. Systemic symptoms range from a non-pruritic rash, arthralgias, myalgias, and adenopathy to complications of the heart and central nervous system. The primary chancre develops in an average of three weeks after exposure; secondary symptoms may occur in about four weeks. The spirochete creates an endarteritis by binding to endothelial cells, which then release a reagin.
Two types of tests for syphilis are available: non-treponemal and treponemal. Because of ease, most screenings are done with rapid plasma regain (RPR), which tests for regain in blood. Alternatively, a venereal disease research laboratory (VDRL) test can be performed. As these do not test for the actual spirochete, and have many false positives, a positive test should be confirmed by a treponemal test, either a fluorescent treponemal antibody–absorbed (FTA–abs) or a microhemagglutination assay–T. pallidum (MHA–TP). Treatment for primary, secondary, and earlylatent syphilis is benzathine penicillin G 2.4 million units IM x 1. Late latent syphilis requires three doses of 2.4 million units IM one week apart. Full treatment and follow-up may require referral. Bicillin® L-A, not Bicillin® C-R, is the recommended product.
Other Considerations
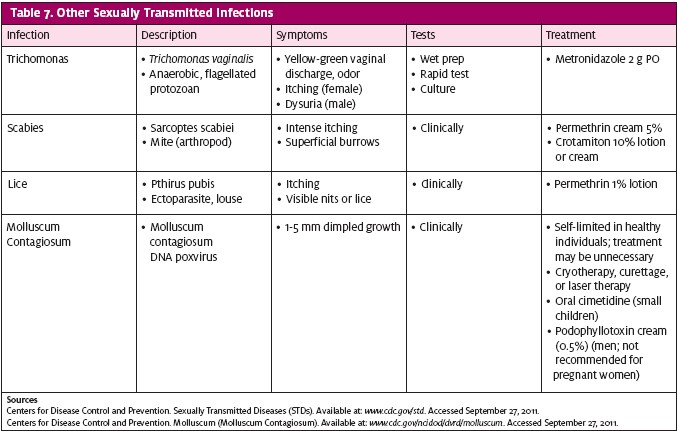
Other infections—trichomonas, scabies, lice, and molluscum contagiosum—can be transmitted from sexual or skin contact (Table 7). If indicated, emergency contraception should be addressed.
Conclusion
Many patients with an STD/I are likely to show up in urgent care. Unexplained rashes, jaundice, adenopathy, hepatic enlargement, abdominal tenderness, skin ulcerations, genitourinary symptoms, or systemic illness should be investigated. Follow-up instructions should be documented. Educating the patient about STDs is essential.
References
- Centers for Disease Control and Prevention (CDC). Sexually transmitted diseases (STDs). CDC website. Available at: www.cdc.gov/std/general/default.htm. Accessed September 27, 2011.
- Stanley M. Pathology and epidemiology of HPV infection in females. Gynecol Oncol. 2010;117(2 suppl):S5-S10.
- Dunne EF, Unger ER, Sternberg M, et al. Prevalence of HPV infection among females in the United States. JAMA. 2007;297(8):813-819.
- Datta SD, Koutsky LA, Ratelle S, et al. Human papillomavirus infection and cervical cytology in women screened for cervical cancer in the United States, 2003-2005. Ann Intern Med. 2008;148(7):557-559.
- Centers for Disease Control and Prevention (CDC). 2009 Sexually transmitted diseases surveillance. CDC website. Available at: www.cdc.gov/std/stats09/default.htm. Accessed September 27, 2011.
- Jastania R. Geddie WR, Chapman W, Boerner ST. Characteristics of apparently falsenegative digene hybrid capture 2 high-risk HPV DNA testing. Am J Clin Pathol. 2006;125(2):223-228.
- Morbidity and Mortality Weekly Report (MMWR). Seroprevalence of herpes simplex virus type 2 among persons aged 14-49 years–United States, 2005-2008. MMWR. 2010;59(15):456-459.
- Morbidity and Mortality Weekly Report (MMWR). Surveillance Summaries. Surveillance for acute viral hepatitis—United States, 2006. MMWR. 2008;57(SS02);1-24
- Vandelli C, Renzo F, Romanò L, et al. Lack of evidence of sexual transmission of hepatitis C among monogamous couples: results of a 10-year prospective follow-up study. Am J Gastroenterol. 2004;99(5):855-859.
- Morbidity and Mortality Weekly Report (MMWR). HIV surveillance–United States, 1981-2008. MMWR. 2011;60(21); 689-693.
- Morbidity and Mortality Weekly Report (MMWR). Antiretroviral postexposure prophylaxis after sexual, injection-drug use, or other nonoccupational exposure to HIV in the United States. MMWR. 2005;54(RR-2):1-20.
- Howard Brown Health Center: STI Annual Report, 2009. Available at: http://www.howardbrown.org/uploadedFiles/Services_and_Programs/HIV_and_Aids_Services/EpiProfile_ 2009_final05.27.pdf. Accessed October 3, 2011.
- National Institute of Allergy and Infectious Diseases (NIAID). Workshop summary: scientific evidence for condom effectiveness for STD prevention. 2001. NIAID website. Available at: http://www.niaid.nih.gov/about/organization/dmid/documents/condomreport.pdf. Accessed September 27, 2011.
Thomas Sunshine, originally trained as an OB/GYN, now works as a full-time urgent care physician at the Anderson, South Carolina, office for DoctorsCare, providing both urgent and primary care. His last article for JUCM was “Assessing Abnormal Intrauterine Bleeding in the Urgent Care Center” (April 2010).