Published on
Urgent message: Sore throat is a complaint commonly encountered in urgent care. Proper evaluation and understanding and use of appropriate antibiotics will foster better patient care and understanding while limiting antibiotic resistance.
William Gluckman, DO, MBA, FACEP and Jessica Kay, PharmD
Pharyngitis refers to the inflammation or irritation of the pharynx, including the tonsils, and can have many etiologies, including a variety of infections, cancer, allergic reactions, gastroesophageal reflux, or toxic inhalations and ingestions. This article will discuss the infectious causes of pharyngitis, the evaluation methods and evidence based management.
Epidemiology
Pharyngitis is a common presenting complaint in urgent care centers, as well as in other outpatient settings. In 2005, 1.2% of all visits to ambulatory care centers and emergency departments were for the complaint of pharyngitis; of those patients, 79% were seen in primary care offices.1
Approximately 90% of all infectious pharyngitis cases in adults, and 80% in children, are caused by viruses.
A small percentage of cases are idiopathic in nature. The most common bacterial agents include Group A beta hemolytic streptococcus (GAS), Groups C, G, and F streptococcus, A haemo lyti cum, M pneumoniae, C pneumoniae, C diphtheriae, and N gonorrhea.
Some of the commo viruses include rhinovirus, coronavirus, coxsackie A, influenza, and herpes. Cytomegalovirus (CMV) and Epstein-Barr virus (EBV) are causes of mononucleosis.
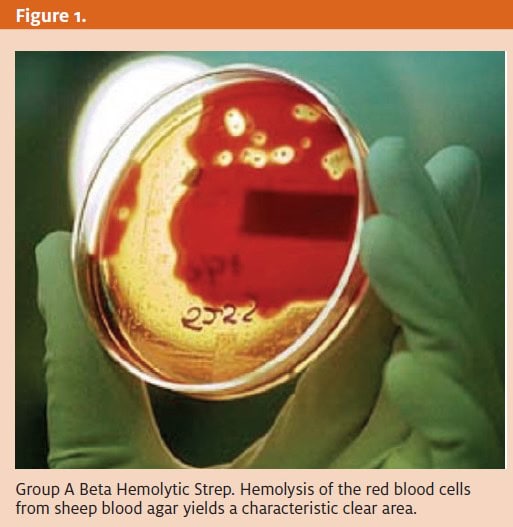
GAS infection (Figure 1) is the most common bacterial cause and follows a seasonal predilection. It is most commonly seen in the winter and early spring. Though adults often suffer from GAS infection, it is most prevalent among children between 5 and 15 years old.
Diagnosis
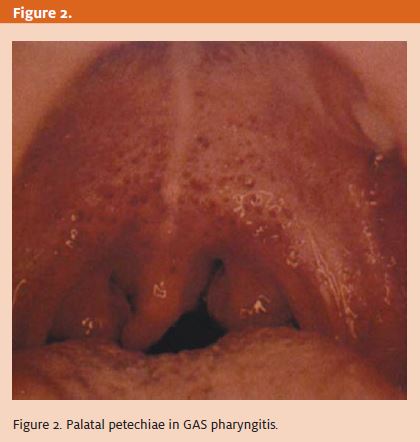
Characteristically, GAS pharyngitis presents with a complaint of acute fever and chills, sore throat, odynophagia, and painful lymphadenopathy in the neck. Headache and nausea/vomiting and abdominal pain also occur and are more common in children. The pharynx will almost universally be erythematous and may or may not have tonsillar exudates. Approximately 10% of cases will have palatal petechiae (Figure 2).
Scarlet fever can occur in the face of GAS infection and produces an erythematous, sand- paper-like rash that begins on the trunk and spreads to the extremities but spares the palms and soles. A “strawberry tongue” may also be present. Tender anterior cervical adenopathy is also common.
However, physical exam findings, overall, are not specific in making a diagnosis of GAS pharyngitis. The presence of tonsillar exudates does not increase the likelihood that GAS is the causative agent; in fact, as noted above, the majority of cases are caused by viruses which also often produce exudates.
Several investigators have developed clinical prediction rules to help determine if the causative agent is GAS, and thus aid in the decision of whether or not to prescribe antibiotics.
Centor looked at presence of tonsilar exudate, swollen or tender anterior cervical nodes, fever history, and absence of cough.2 He found a positive predictive value of only 56% when all four of these were present.
The McIsaac score evaluated similar signs and symptoms and assigned scores based on age and these criteria.3,4 Both were both found to be relatively equivalent.5
Rapid antigen detection testing (RADT) for GAS is commonly performed in urgent care and other similar settings, and has a high degree of sensitivity (80% to 90%) and specificity (³ 95%),6,7 making this a valuable tool the urgent care practitioner. Infectious Diseases Society of America (IDSA) guidelines support the use of RADT in all suspected cases of GAS and, because of the high specificity, negative results do not warrant follow-up throat culture to confirm a true vs. false negative result.
In children and adolescents, a culture is suggested unless the clinician has shown that the RADT has demonstrated comparable results to cultures in that specific practice.
Complications
GAS pharyngitis may lead to one or more complications:
- Suppurative complications:
- Peritonsillar abscess (quinsy)
- Retropharyngeal abscess
- Cervical lymphadenitis
- Non-suppurative complications:
- Scarlet fever
- Rheumatic fever
- Acute post-streptococcal glomerulonephritis (APSGN)
Pitfalls
One common mistake when evaluating the patient complaining of a sore throat is to not examine the throat fully.
A peritonsillar abscess will often present with the same symptoms; however, a careful examination of the pharynx will reveal a swelling medial to the tonsil and deviation of the uvula to the unaffected side. These patients also tend to have trismus and often appear toxic.
Treatment
The goals of treatment of pharyngitis are to limit the suppurative and nonsuppurative complications and decrease the duration of clinical signs and symptoms. Improving patient comfort and decreasing the incidence of adverse drug reactions are also important.
Early antibiotic treatment of streptococcal pharyngitis may lead to earlier resolution of symptoms and shorten the course of illness by about one day, but can increase risk of resistance and recurrence and may decrease immune response.
It is thought that patients no longer transmit GAS pharyngitis after 24 hours of antibiotic treatment. Microbiological elimination with antibiotics usually occurs within 48 to 72 hours.8,9
Although early treatment decreases the risk of transmission, data suggest that therapy may be delayed for two to three days (up to a maximum of nine days) after the on- set of symptoms and still prevent the occurrence of complications. This approach is particularly useful in patients with frequent, recurring, mild-to-moderate infections.
Linder in 2005 reported that 53% of children with sore throat received antibiotics. Antimicrobial therapy should be limited to those who have clinical and epidemiologic features of GAS pharyngitis with a positive laboratory test.10 The authors agree with the IDSA that recommended treatment should be based on clinical criteria and positive rapid streptococcal antigen test (RSAT) or culture results in order to diagnose GAS.
Clinical decision rules have been shown to decrease antibiotic prescription writing. These recommendations are of importance to prevent the inappropriate use of antibiotic therapy.
Pharmacologic Therapy
Analgesics
Systemic analgesics/antipyretics are recommended for pain relief. Acetaminophen is preferred due to concerns over NSAIDs increasing the risk for developing necrotizing fascitis/toxic shock syndrome, which has been associated with GAS infections. Topical analgesics such as viscous lidocaine and lozenges, along with other non-pharmacologic supportive care such as rest, fluids, and salt water gargles may resolve symptoms up to one to two days faster.9
The value of good analgesia should not be underestimated. Patients seek care mostly to make them feel better—i.e., pain relief. Many will ask for antibiotic prescriptions thinking that this is the best and fastest route to resolution of the problem. Urgent care providers can send patients home without an antibiotic when it isn’t needed and still achieve high lev- els of patient satisfaction if the patient’s pain is addressed adequately.
Antibiotics
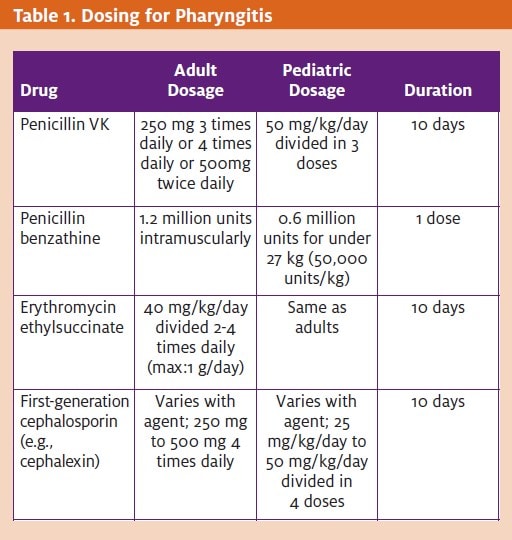
Antibiotic therapy has been the mainstay treatment for GAS pharyngitis. The primary treatment options consist of penicillins (pri- mary treatment), cephalosporins, macrolides, and clindamycin (Table 1).
In patients allergic to penicillins, a macrolide should be used. A first-generation cephalosporin may be used if the penicillin reaction is a non-IgE mediated hypersensitivity reaction.
- Penicillins—Interfere with bacterial cell wall syn- thesis by inhibiting the formation of peptidoglycan crosslinks during active multiplication, causing cell wall death and resultant bactericidal activity against susceptible Penicillins are currently recom- mended as the antimicrobial agent of choice for the treatment of GAS pharyngitis.
This recommendation is based upon its accept- able safety and efficacy in eradicating infection, its narrow spectrum of activity, and its economical cost.6 Although surprisingly in 2001 Kaplan showed resistance rates for benzathine penicillin G IM and oral penicillin V to be 37% and 35%, respectively, in pediatric patients, it remains the recommended treatment.11 Usual duration of therapy to prevent further systemic complications is 10 days. Gastrointestinal issues and rash are the most common side effects.
Benzathine penicillin G—Patients who may not be willing or able to comply with a 10-day course of therapy may be given a single dose of benzathine penicillin G 1.2 million units IM.
Though amoxicillin has a more extended spectrum of coverage for pathogens than penicillin VK, its use may increase compliance in children because it is more palatable in the suspension form.
- Cephalosporins—Inhibit bacterial cell wall synthesis by binding to one or more of the penicillin-binding proteins, which in turn inhibits the final transpeptidation step of peptidoglycan synthesis in bacterial cell walls, thus inhibiting cell wall biosynthesis.
Cephalosporins (e.g., cefpodoxime, cefdinir) may be more effective and have better eradication rates after a five-day therapy compared with a 10-day therapy with penicillins.
- Macrolides—Bind to the 50s ribosomal subunit, resulting in blockage of transpeptidation, which inhibits RNA- dependent protein synthesis at the chain elongation
Macrolide antibiotics (e.g., erythromy- cin, clarithromycin, azithromycin) are the drugs of choice in patients who are allergic to penicillin. Newer macrolides such as azithromycin and clarithromycin are as effective as erythromycin and cause fewer gastrointestinal side effects. Cholestatic hepatitis may occur mainly in pregnant adult patients receiving erythromycin estolate. Resistance rates are low at approximately <5%.12 Erythromycin estolate and ethylsuccinate are more comparable to oral penicillin for eliminating GAS pharyngitis than erythromycin base or stearate. Azithromycin and clarithromycin are safe, require only five days of therapy, and are as effective as both penicillin and erythromycin. These medications should only be used in patients not responding to penicillin or who are unable to tolerate either penicillin or erythromycin.
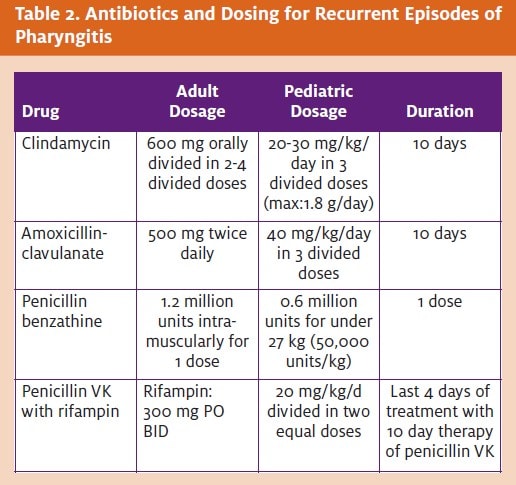
- Clindamycin—Reversibly binds to 50s ribosomal subunits, preventing peptide bond formation, thus inhibiting bacterial protein Clindamycin is bacteriostatic or bactericidal, depending on drug concentration, infection site, and organism. It can be used in patients who are penicillin-allergic, and also as an alternative for macrolide resistance. Due to its potential to cause pseudomembranous colitis, it is recommended in patients with multiple, recurrent episodes of GAS pharyngitis or allergies to both penicillins and erythromycins.
In patients with recurrent episodes of GAS pharyngitis, treatment should include [3-lactamase-resistant antibiotics against aerobic and anaerobic organisms (Table 2). It should consist of clindamycin or amoxicillin-clavulanate due to the high rates or eradication.
Resistance
Penicillin is currently recommended as first-line ther- apy. Erythromycin is the recommended alternative in penicillin-allergic patients. First-generation cephalo- sporins can also be used as an alternative.
Due to increased use of broad-spectrum antibiotics, such as newer macrolides, second- and third-generation cephalosporins, and amoxicillin-clavulanate, problematic increases in resistance among the respiratory pathogens have been seen, and thus their routine or first-line use has not been recommended.
Many cases have been reported in which penicillin failed to eliminate group A streptococcus from “GAS carriers.” One study, designed to evaluate the potential of various antibiotics to eliminate Group A streptococcus, found that GAS continued to exist regardless of treating it with penicillin.12 GAS was eliminated when treated with erythromycin or azithromycin.
Cephalothin (a cephalosporin), and clindamycin were more effective in killing GAS than penicillin, but were also less effective than erythromycin or azithromycin. It was concluded that failure to eliminate GAS was due to a lack of effective penicillin entry into the epithelial cells. Al- though resistance rates remain higher than with other available treatments, it is still preferred as first-line therapy due to its narrow spectrum of activity and extremely low cost. Macrolide resistance in the United States is low (<5%) and not widespread, whereas in areas such as Japan and Finland increased resistance remains an issue. How- ever, there have been reports of outbreak of macrolide resistant GAS pharyngitis in the United States. Resistance may be a concern if these agents are routinely overused. GAS resistance rates to tetracyclines and sulfonamides are high; therefore, use of these agents is no longer recommended.6,12,13
Viral Pharyngitis
The use of corticosteroids remains controversial, but has been shown to decrease pain and shorten the duration of symptoms without increasing complications. Corticos- teroids (e.g., dexamethasone, prednisone) may be used in patients who are symptomatic and have compromised airways.
In patients with viral pharyngitis, supportive care is recommended. In patients who are immunocompromised, antivirals may have some clinical benefit. In severe cases of herpes simplex pharyngitis and for immunocompromised patients, acyclovir, famciclovir, and valacyclovir are recommended. In CMV infections in immunocompromised patients, foscarnet or ganciclovir is recommended. In patients with oral thrush, antifungals (nystatin, flu-conazole) may also be used.
Summary
Differentiation of bacterial pharyngitis from other causes poses some clinical challenges. Through a combination of history, physical exam findings, clinical predictive rules, and rapid strep antigen testing, most cases requiring antibiotic treatment can be identified, and inappropriate antibiotic administration can be avoided.
Penicillin remains the drug of choice in treating GAS pharyngitis, and there continues to be several alterna- tives for treatment failures due to allergy and resistance. Consideration to maximizing patient comfort with lib- eral analgesic—along with judicious antibiotic—use will improve patient satisfaction and help decrease antibiotic resistance. ■
REFERENCES
- Burt CW, McCaig LF, Rechtsteiner Ambulatory Medical Care Utilization Estimates for 2005. Advance Data from Vital and Health Statistics; no 388. Hyattsville, MD: Na- tional Center for Health Statistics. 2007. Available at: www.cdc.gov/nchs/data/ad/ad388.pdf.
- Centor RM, Witherspoon JM, Dalton HP, et The diagnosis of strep throat in adults in the emergency room. Med Decision Making. 1981;1:239-246.
- McIsaac WJ, Goel V, To T, et The validity of a sore throat score in family practice. CMAJ. 2000;163:811-815.
- McIsaac A clinical score to reduce unnecessary antibiotic use in patients with sore throat. CMAJ. 1998; 158:75-83.
- Schwartz K, Monsur, Joseph, et Pharyngitis clinical prediction rules: Effect of in- terobserver agreement. A MetroNet Study. J Clin Epidemiol. 2004; 57:142-146
- Bisno AL, Gerber MA, Gwaltney JM, et Practice guidelines for the diagnosis and management of group A streptococcal pharyngitis. Clin Infect Dis. 2002;35(2):113-125.
- Gerber Comparison of throat cultures and rapid strep tests for diagnosis of strep- tococcal pharyngitis. Pediatr Infect Dis J. 1989;8:820-824.
- Ebell M, Smith M, Barry H, et al. Does this patient have step throat? JAMA. 2000;284:2914-2918.
- Snow V, Mottur-Pilson C, Cooper RJ, et al, for the American College of Physicians, Amer- ican Society of Internal Medicine. Principles of appropriate antibiotic use for acute pharyngitis in Clinical practice guideline, part I. Ann Intern Med. 2001;134;506-508
- Linder JA, Bates DW, Lee GM, et Antibiotic treatment of children with sore throat. JAMA. 2005;294(18):2315-2322.
- Kaplan EL, Johnson Unexplained reduced microbiological efficacy of intramus- cular benzathine penicillin G and or oral penicillin V in eradication of Group A strepto- cocci from children with acute pharyngitis. Pediatrics. 2001;108:1180-1186.
- Kaplan EL, Johnson DR, Del Rosario MC, et Susceptibility of group A beta-hemolytic streptococci to thirteen antibiotics: Examination of 301 strains isolated in the United States between 1994 and 1997. Pediatr Infect Dis J. 1999;18:1069-1072.
- Seppala H, Klaukka T, Vuopio-Varkila J, et The effect of changes in the consump- tion of macrolide antibiotics on erythromycin resistance in group A streptococci in Fin- land. N Engl J Med. 1997;337:441-446.
SUGGESTED READINGS
- Bisno Acute pharyngitis. N Engl J Med. 2001;344(3):205-211.
- Bisno Are cephalosporins superior to penicillin for treatment of acute streptococ- cal pharyngitis? Clin Infect Dis. 2004;38:1535-1537.
- Bisno AL, Gerber MA, Gwaltney JR et Diagnosis and management of group A strep- tococcal pharyngitis: A practice guideline. Clin Infect Dis. 1997;25(3):574-583.
- Bulloch B, Kabani A, Tenenbein Oral dexamethasone for the treatment of pain in chil- dren with acute pharyngitis: a randomized, double-blind, placebo-controlled trial. Ann Emerg Med. 2003;41(5):601-608.
- Carroll KC, Monroe P, Cohen S et Susceptibility of beta-hemolytic streptococci to nine antimicrobial agents among four medical centers in Salt Lake City, Utah, USA. Diagn Mi- crobiol Infect Dis. 1997;27:123-128.
- Casey JR, Pichichero Meta-analysis of cephalosporins versus penicillin for treatment of group A streptococcal tonsillopharyngitis in adults. Clin Infect Dis. 2004; 38:1526-1534.
- Casey JR, Pichichero Meta-analysis of cephalosporin versus penicillin treatment of group A streptococcal tonsillopharyngitis in children. Pediatrics. 2004;113:866-882.
- Cohen R, Levy C, Doit C et Six-day amoxicillin vs. ten-day penicillin V therapy for group A streptococcal tonsillopharyngitis. Pediatr Infect Dis J. 1996;15:678-682.
- Cooper RJ, Hoffman JR, Bartlett JG et Principles of appropriate antibiotic use for acute pharyngitis in adults: Background. Ann Intern Med. 2001;134(6):509-517.
- Cooper RJ, Hoffman JR, Bartlett JG et Principles of appropriate antibiotic use for acute pharyngitis in adults: Background. Clinical Practice guideline. Ann Emerg Med. 2001;37(6):711-719.
- Del Mar CB, Glasziou PP, Spinks Antibiotics for sore throat. Cochrane Database Syst Rev. 2006. Oct 18;(4):CD000023.
- Gerber Diagnosis and treatment of pharyngitis in children. Pediatr Clin N Am. 2005;52:729-747.
- Jacobs JA, deKrom MC, Kellens JT et Meningitis and sepsis due to group G strepto- coccus. Eur J Clin Microbiol Infect Dis. 1993;12:224-225.
- Kaplan EL, Chhatwal GS, Rohde Reduced ability of penicillin to eradicate ingested group A streptococci from epithelial cells: clinical and pathogenetic implications. Clin In- fect Dis. 2006;43(11):1398-1406.
- Lacey CF, Armstrong LL, Goldman MP et Drug Information Handbook. 15thEd. Hud- son, Ohio. Lexi-Comp Inc. 2008.
- Marvez-Valls EG, Stuckey A, Ernst A randomized clinical trial of oral versus intramus- cular delivery of steroids in acute exudative pharyngitis. Acad Emerg Med. 2002;9(1):9- 14.
- McMillan JA, Weiner LB, Higgins AM et Pharyngitis associated with herpes simplex virus in college students. Pediatr Infect Dis J. 1993;12(4):280-284.
- Peyramond D, Portier H, Geslin P et 6-day amoxicillin versus 10-day penicillin V for group A beta-haemolytic streptococcal acute tonsillitis in adults: A French multicentre, open-label, randomized study. The French Study Group Clamorange. Scand J Infect Dis. 1996;28(5):497-501.
- Pichichero Cephalosporins are superior to penicillin for treatment of streptococcal tonsillopharyngitis: Is the difference worth it? Pediatr Infect Dis J. 1993;12:268-274.
- Pichichero ME, Cohen Shortened course of antibiotic therapy for acute otitis media, sinusitis, and tonsillopharyngitis. Pediatr Infect Dis J. 1997;16(7):680-695.