Published on
Urgent message: Being able to recognize the distinct oral lesions of common illnesses in children is essential, but it can be difficult to conduct an oral examination in frightened young children.
Introduction
Inspecting intraoral lesions in children will often confirm a diagnosis, but getting uncooperative patients to let the clinician visualize such lesions is challenging. Here we provide helpful examination tips and review common pediatric infectious and allergic oral lesions and their treatment.
Techniques for Examination of the Oral Cavity in Children
To many children, oral examinations are frightening. Improper immobilization results in an inadequate examination, increased patient distress, and increased potential for injury to both the child and the healthcare provider. Tips for performing an adequate and safe examination include the following:
- The clinician should wear a mask and eye protection as safeguards against spitting or coughing by the patient. A tongue blade, gauze, an adequate light source, and specimen-collection devices should be easily accessible.
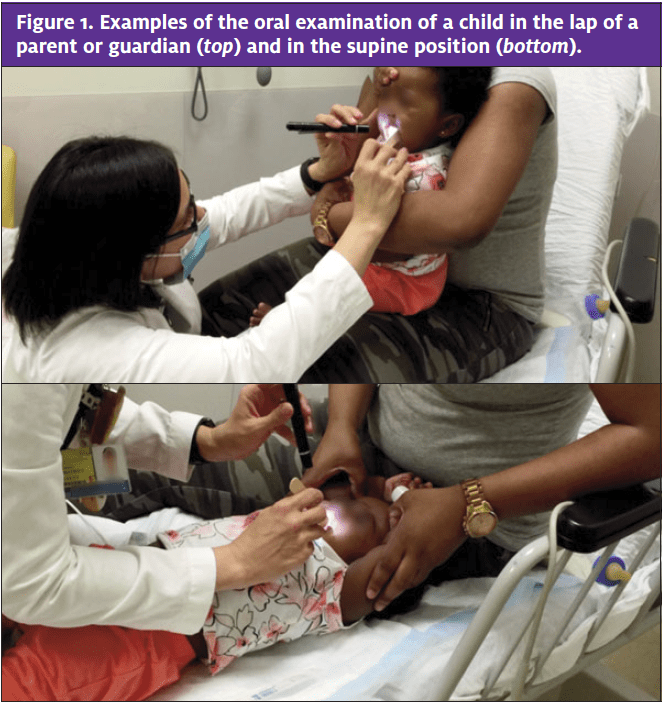
- Children who can sit upright may sit in their parent’s or guardian’s lap, whereas children of any age can lie supine on the examination table (Figure 1).
• For the lap examination, seat the child facing sideways or facing the clinician. Have the parent or guardian wrap one arm across the child, encompassing both arms of the child with that
arm, and use their free hand to immobilize the child’s head against their chest. A child who is kicking can be immobilized if the parent or guardian crosses one leg over the child’s legs.
• For examining a child in the supine position, lay the child on the examination table. The parent or guardian holds both arms of the child alongside the child’s ears, with lateral pressure at
the upper arm to limit side-to-side movements of the child’s head. The clinician may lean over the child’s torso to limit leg or trunk movements during the examination. - Once the child is immobilized, the mouth can be examined. A tongue blade causes the least gagging or biting when inserted vertically along the inside of the cheek until it reaches the posterior buccal shelf. A vertical tongue blade placed at the gap between clamped teeth and the cheek may encourage mouth-opening by limiting the child’s ability to close the mouth completely. When the child cries, the clinician can swiftly shift the blade to the center of the tongue to see the posterior oropharynx. For inspecting the gingiva, buccal mucosa, and tongue, the provider should use gauze for grasping wet mucosal tissue.
Hand-Foot-and-Mouth Disease
Hand-foot-and-mouth disease (HFMD) is a common cause of oral exanthems in young children during the summer and fall. Caused by coxsackievirus, an enterovirus, HFMD is highly contagious and spreads through respiratory transmission or through contact with vesicles and stool.1
Medical History and Physical Examination
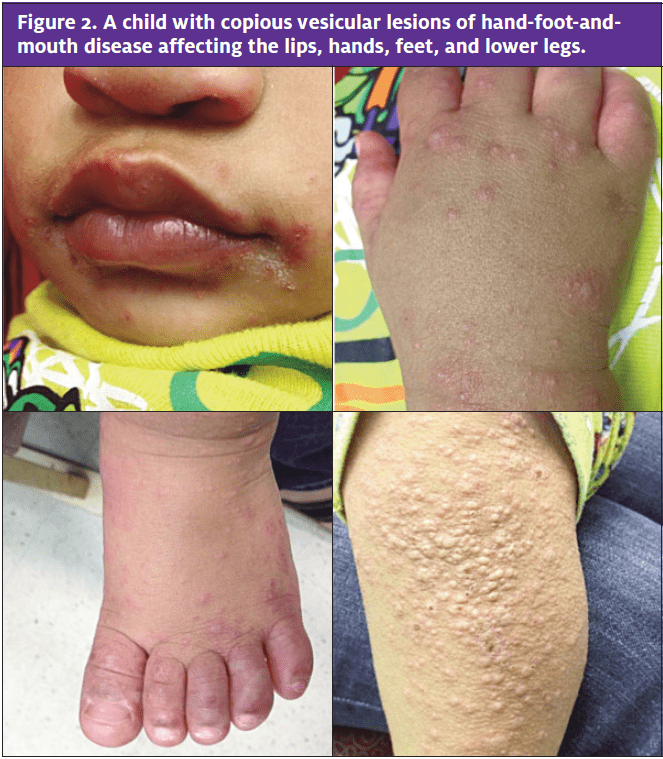
The classic presentation is a prodrome of fever and malaise, followed by development of painful oral ulcerations of the palate, buccal mucosa, tongue, or lips and an erythematous maculopapular or vesicular rash on the palms, soles, extremities, buttocks, or trunk. Concurrent respiratory (e.g., cough, rhinorrhea) or gastrointestinal (e.g., vomiting, diarrhea) symptoms may be present. Verbal children report a sore throat, whereas nonverbal children may present with decreased oral intake, fussiness with oral feedings, increased drooling, or irritability (Figure 2).
Diagnosis
The diagnosis of HFMD is made by findings on the clinical examination and medical history. Viral culture is not necessary unless atypical symptoms are present or if other etiologies (e.g., herpes simplex virus [HSV]) are suspected.
Red Flags
Rare, severe cases of HFMD can occur with the development of viral meningitis and encephalitis. Recently, atypical presentations of HFMD have been seen worldwide, presenting with extensive cutaneous involvement and attributed to other enteroviral species.2
Treatment
Treatment for HFMD is supportive because the disease is self-limited, lasting 7 to 10 days. The most common clinical challenge in HFMD is pain control and hydration, particularly in younger children. Oral analgesics are often adequate for pain control. A commonly used, though not well-studied, topical oral analgesic is “magic mouthwash”: a mixture of liquid diphenhydramine, Maalox, and viscous lidocaine 2% at a 1:1:0.5 ratio. Dosing is limited by the diphenhydramine at 5 mg/kg per day every 6 hours. Include the lidocaine only if the patient is able to swish and spit.3 Encourage small, frequent sips of an oral rehydration solution, such as Pedialyte, if the child has decreased oral intake. Consider emergency department transfer if the child has moderate to severe dehydration and requires intravenous fluids.
Herpetic Gingivostomatitis
Herpetic gingivostomatitis is a common cause of oral lesions in young children who have been infected with HSV. Transmission is generally through saliva or direct contact with infected lesions.
Medical History and Physical Examination
Primary outbreaks in children are more severe than subsequent outbreaks, with development of vesicular lesions over the lips, tongue, gingiva, buccal mucosa, and palate. These lesions are very painful and can contribute to poor oral intake, particularly in the younger child. The presentation of the oral vesicles during a primary outbreak is often heralded by fever, generalized malaise, lymphadenopathy, and friable, erythematous gingiva. Subsequent outbreaks are less severe, with lesions localized to the lips. Close contacts and family members are often the source of transmission and may report oral lesions.
Diagnosis
Diagnosis is made clinically, by confirmation of vesicles at different stages of healing on oral mucosa or the lips. Diagnostic testing with viral culture, serology, or polymerase chain reaction may be used if the etiology must be confirmed.4
Red Flags
Children can autoinoculate themselves with HSV at other sites. Be sure to evaluate the fingers of children who engage in thumb-sucking for herpetic whitlow or eczema herpeticum. Lesions near or involving the eye are at risk of HSV epithelial keratitis, which can result in scarring and permanent vision loss.5 Ophthalmologic consultation is warranted because of the associated morbidity
and potential for recurrence.
Treatment
Systematic review of oral acyclovir for primary herpetic gingivostomatitis in children younger than 6 years of age shows weak evidence that it will decrease the number of oral lesions, the formation of new lesions, oral intolerance, or the rate of hospital admission.6,7 Despite limited evidence, we recommend that clinicians prescribe oral acyclovir within 3 to 4 days of disease onset,
and for those with severe pain or difficulty maintaining hydration. Supportive management of herpetic gingivostomatitis includes oral analgesics for pain control and oral hydration. Barrier ointments (e.g., petroleum jelly) may be soothing and prevent lip adhesion.
Streptococcal Pharyngitis
Group A -hemolytic streptococcal (GAS) pharyngitis, commonly called strep throat, is the most common type of bacterial pharyngitis in children. GAS pharyngitis is most commonly seen in children between the ages of 5 and 15 years and tends to occur between late fall and early spring.
Medical History and Physical Examination
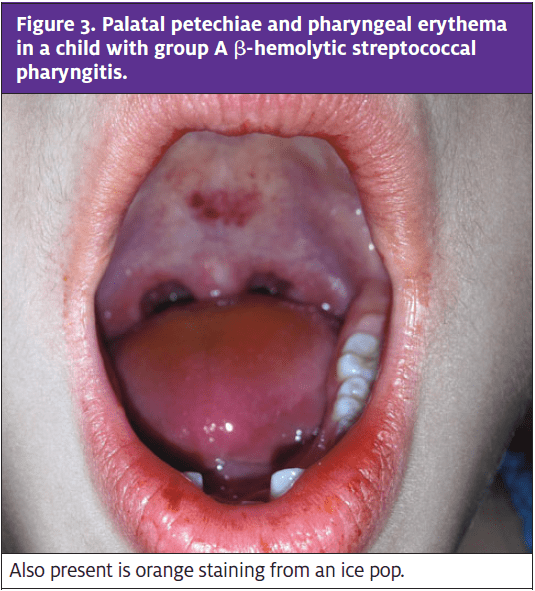
Classic signs of GAS pharyngitis include a sudden onset of pain with swallowing, fever, headache, abdominal pain, nausea, and vomiting, with an absence of coughing, conjunctivitis, or other systemic symptoms. Intraoral findings include enlarged, erythematous, exudative tonsils and palatal petechiae (Figure 3). Enlarged, tender cervical lymph nodes or a fine, sandpaper-like scarlatiniform rash may be present.
Diagnosis
Clinical findings alone are not adequate to diagnose GAS pharyngitis, and dependence on them leads to inappropriate antibiotic use. Throat culture and rapid antigen detection test are the gold standards for diagnosis. Because the incidence of GAS pharyngitis is greater in children than in adults, obtaining a throat culture is recommended if findings on the antigen test are negative.
Children should be tested on the basis of their risk factors for GAS pharyngitis, which include age (5–15 years), seasonality, and clinical signs and symptoms. Routine testing for GAS pharyngitis is generally not indicated in children younger than 3 years of age because of the low incidence of the disease in this age group and the low likelihood of complications.8
A number of clinical decision rules (e.g., modified Centor criteria) have been evaluated for their ability to predict the likelihood that findings on throat culture will be positive for GAS pharyngitis; however, metaanalysis suggests poor specificity for the rules. Current recommendations are to use rapid testing for diagnosis rather than rely on clinical decision rules alone.9
Differential Diagnosis
Additional clinical findings may suggest other diagnoses (e.g., abscess, ulcers) or may reveal symptoms of other entities (e.g., rhinorrhea, cough, and conjunctivitis).8 Viral pathogens (e.g., adenovirus, influenza, entero – viruses) are a common cause of pharyngitis. Epstein-Barr virus (EBV) and cytomegalovirus, the viruses causing mononucleosis syndrome, similarly produce fever,
exudative tonsils, and lymphadenopathy.
Red Flags
Lemierre syndrome, septic thrombophlebitis of the internal jugular vein, should be considered if a patient with GAS pharyngitis appears septic and has unilateral swelling or tenderness of the throat and neck. The infection occurs from contiguous spread of an oropharyngeal infection.
Treatment
Although GAS pharyngitis is a self-limited condition, treatment shortens the duration of symptoms, decreases transmission, and prevents development of acute rheumatic fever.5 The first-line treatments include amoxicillin and a single dose of intramuscular penicillin G benzathine. Hydration and pain control are tenets of symptomatic management. Steroids are not routinely recommended for children and adolescents with pharyngitis, because of insufficient evidence of benefit.10
Mononucleosis
Infectious mononucleosis is a viral illness most often caused by EBV. Often called the kissing disease, it is most common during adolescence and preadolescence and is transmitted through respiratory secretions. By adulthood, most people are EBV-seropositive after subclinical or symptomatic infections.
Medical History and Physical Examination
Mononucleosis presents as pharyngitis, anterior and posterior cervical lymphadenopathy, fever, and fatigue. Its symptoms and clinical presentation can mimic those of GAS pharyngitis, including pharyngeal exudate, erythematous tonsils, and palatal petechiae, but with a several-day prodrome of fatigue and anorexia.
Diagnosis
The severity of pharyngitis is generally less than that of GAS pharyngitis, but fever and malaise may remain prolonged, lasting even several weeks. A prolonged duration of symptoms should lead the clinician to consider mononucleosis instead of GAS pharyngitis, or even coinfection by mononucleosis and GAS pharyngitis, despite negative findings on throat culture for GAS. Although the diagnosis is generally a clinical one, the presence of >10% atypical lymphocytes is suggestive of mononucleosis, and confirmation can be obtained through a mononucleosis spot test (also called a
monospot test) or EBV titers. A monospot may produce a false negative result early in the course.
Red Flags
A potentially dangerous manifestation of infectious mononucleosis is hepatosplenomegaly, which can predispose active children and adolescents to splenic rupture. Rupture can be life-threatening, so contact sports must be avoided until splenomegaly is resolved, or 4 weeks from diagnosis. Four weeks has been chosen by consensus as a safe point for return to sports, though
evidence is lacking. For athletes desiring to participate sooner or for those with equivocal examination findings, spleen size may be evaluated by abdominal ultrasonography. 11
Treatment
Patients with hepatosplenomegaly are advised to avoid activity. Treatment is supportive, with hydration, rest, and oral analgesia. There is limited evidence to support the use of steroids with or without the use of antivirals for symptomatic relief.12
Peritonsillar Abscess
A peritonsillar abscess (PTA) is the collection of pus between the capsule of the tonsil and the pharyngeal muscles. It often arises from contiguous spread of a pharyngeal infection.
Medical History and Physical Examination
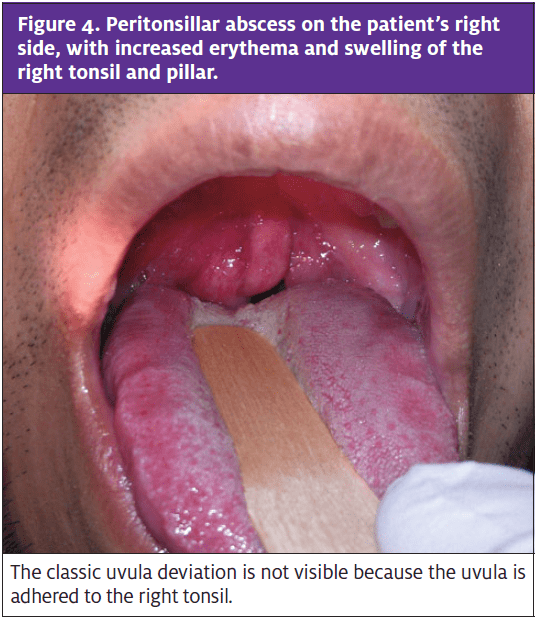
The features of a PTA often encountered in the medical history are fever, (unilateral) throat pain, and a muffled or “hot potato” voice. There may be a history of a preceding upper respiratory infection. Patients are typically adolescents. They have difficulty swallowing, drool, and cannot tolerate oral intake. Physical examination reveals a fluctuant bulge with erythema and surrounding edema at the tonsil or pillar unilaterally. There may also be asymmetry of the palate, uvula deviation, trismus, and cervical or submandibular lymphadenopathy (Figure 4). The presentation of peritonsillar cellulitis is similar, but without fluctuance, uvula deviation, and trismus.
Diagnosis
Diagnosis is often made clinically, and it is confirmed on release of pus after drainage. Laboratory work (complete blood cell count, C-reactive protein, metabolic panel, or blood cultures) may be helpful if there is the possibility of systemic infection or dehydration. A rapid antigen detection test is helpful because Group A -hemolytic streptococcus is a frequent bacterial pathogen. However, PTA infections are often polymicrobial, with mixed aerobic and anaerobic bacteria.13
Radiologic evaluation with ultrasonography or computed tomography is indicated when there is the possibility of advanced parapharyngeal infection, or when it is necessary to differentiate abscess from cellulitis.14 Lateral neck x-rays are not indicated for this diagnosis, though they are helpful in the young child with retropharyngeal abscess (RPA).
A PTA may be indistinguishable from peritonsillar cellulitis until drainage is performed to confirm the presence of pus. A bacterial culture is indicated for immunocompromised patients who are at risk of infection by resistant organisms, but it is not recommended for routine analysis, because culture findings often do not alter treatment.14
Differential Diagnosis
Viral or bacterial pharyngitis may present with similar symptoms. Consider infectious mononucleosis as a concurrent or alternative diagnosis.14 The features that distinguish PTA from pharyngitis are the asymmetry seen in the palate and tonsillar pillars. Some clinicians consider RPA when thinking about infections of the deep neck space, but this is a separate entity. RPA presents in toddlers to preschool-age children with fever, irritability, drooling or difficulty handling secretions, and neck stiffness. On examination, the child may tripod or have discomfort
moving the neck. Trismus, muffled voice, or visible posterior pharyngeal changes can be present in RPA.
Red Flags
Watch for the child who is systemically ill, is having respiratory difficulty, or is dehydrated from poor oral intake. Complications of PTA include airway obstruction, rupture of the abscess with aspiration of pus, and extension to soft issues, causing an infection of the deep neck space.14 Consider Lemierre syndrome as a complication of a PTA.
Treatment
Initial treatment in the urgent care setting includes confirming a patent airway and controlling pain and fever. If the patient appears systemically ill, provide intravenous hydration. If the provider is confident that this is a peritonsillar cellulitis, a 24-hour trial of oral antibiotics with close follow-up is an option, but it is important to give the patient’s parent or guardian clear instructions to seek care for the child if there is no improvement (i.e., if there is persistent fever, throat pain or swelling, or difficulty breathing). If there is the possibility of PTA, the patient should be referred to a hospital with otorhinolaryngology coverage to perform needle aspiration or incision and drainage. A 14-day course of antibiotics to cover oral flora is indicated for PTA or cellulitis. Parenteral recommendations are clindamycin or amoxicillin-sulbactam, and oral options are clindamycin or amoxicillin-clavulanate.
Thrush
Thrush is a fungal infection of the mouth, most commonly caused by Candida albicans. It occurs in healthy infants, but immunocompromised patients, those with diabetes mellitus, and those taking inhaled cortico – steroids are also at risk.
Medical History and Physical Examination
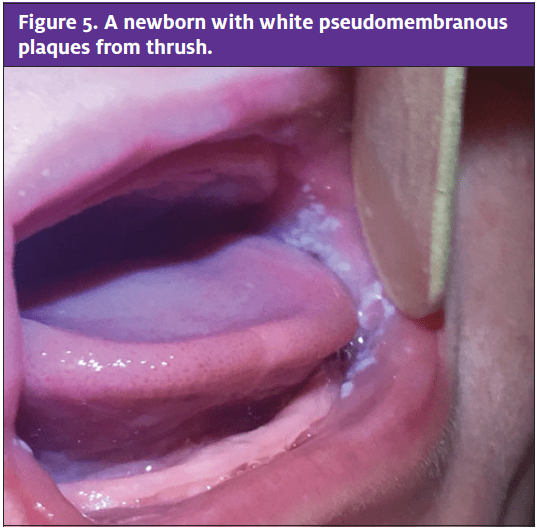
Thrush can be easily missed in newborns because infants are often asymptomatic. The symptomatic infant may have difficulty feeding. Concomitant findings may include a diaper rash in the infant or increased nipple pain and redness in the breast-feeding mother. The provider should inspect the buccal mucosa, palate, and tongue using a tongue depressor to raise the cheek or open the mouth. Examination findings include a pseudomembranous white plaque on any part of the oral cavity; the plaque does not scrape off with a tongue blade (Figure 5). If the infant has a diaper rash from Candida, there may be erythematous patches or papules (i.e., satellite lesions), with areas of white scale, in the diaper area. The breast-feeding mother may have erythematous,
shiny, flaky, or very painful nipples.
In an infant with a chief symptom of diaper rash, check the mouth for associated thrush, and vice versa. Oral candidiasis can also present as angular cheilitis after frequent licking at the corners of the mouth.
Diagnosis
Thrush is a clinical diagnosis. White plaques in the mouth are distinguishable from milk because milk will scrape off the mucosa, whereas thrush will not. Microscopic confirmation can be made with a potassium hydroxide preparation.
Red Flags
In an otherwise healthy child older than 1 year of age with thrush, consider underlying immunologic or endocrinologic disorders, such as human immunodeficiency virus, a primary immunodeficiency, or diabetes mellitus.
Treatment
The infant should be prescribed oral nystatin, and the parent or guardian should be given instructions to give the medication until the lesions are resolved (typically within 2 weeks), plus an additional 3 days.15 Fluconazole is an alternative if there is treatment failure with nystatin. 16 Diaper rash and nipple candidiasis are treated with a topical antifungal cream or ointment, such as
nystatin or clotrimazole, used after each diaper change or feeding, respectively. Topical antifungals should be applied for the same duration as the oral medicine. Advise the mother to apply the topical antifungal onto her nipple just after breast-feeding the infant, to limit ingestion of the topical formulation.17
Parents must be educated on the ubiquitous nature of Candida and the possibility of reinfection if all fomites are not cleaned. All artificial nipples (including those used in pacifiers and bottles) must be boiled or cleaned with hot, soapy water after each use. Emphasize the importance of treating a breast-feeding mother’s nipples because they are a source of shared Candida infection.
The parent or guardian should follow up with the child’s pediatrician in 1 month if the symptoms are not resolved.
Allergic Rhinitis
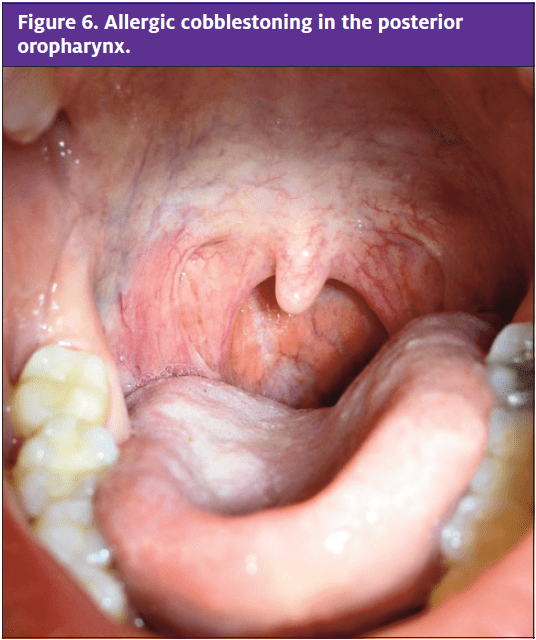
Allergic rhinitis frequently leads to a sore throat because of cobblestoning or lymphoid hyperplasia in the posterior oropharynx (Figure 6).
Medical History and Physical Examination
When there is an allergic etiology, the clinician may elicit a history of rhinorrhea, nasal congestion or obstruction, cough, fatigue, or pruritus of the eyes, nose, and palate. Patients with rhinitis often wake up with a sore throat. On examination, the provider may find that the posterior oropharynx has cobblestoning, the turbinates are boggy, and the conjunctiva is injected. The source of sore throat and cobblestoning is a combination of postnasal drip and mucus accumulation in the throat, leading to hyperplastic pharyngeal lymphoid tissue. Other examination findings include a horizontal fold at the nasal bridge (allergic salute) and infraorbital edema or discoloration (allergic shiners).18
Diagnosis
This is a clinical diagnosis based on medical history and inspection of the posterior oropharynx.
Red Flags
Other causes of cobblestoning include rhinitis due to viral infection, sinusitis, gastroesophageal reflux disease, and, rarely, inflammatory bowel disease.19 If antihistamines or nasal corticosteroids are not effective, consider an alternative source of throat pain with cobblestoning. Allergic rhinitis develops only after years of allergen exposure. If the child is younger than 2 years of age, consider alternative diagnoses.
Treatment
Mild, intermittent allergic rhinitis is treated with an oral antihistamine, such as cetirizine or loratadine. Add a nasal corticosteroid for moderate or persistent symptoms. 18,20 Remind the child’s parent or guardian that nasal steroids can provide immediate symptomatic relief but that full benefit may take days to weeks.18 If the source appears to be viral or infectious, nasal steroids
will not help in the short time frame of symptomatology. Recommend supportive care, or a spoonful of honey to help with cough and sore throat (if the child is older than 1 year of age). The American Academy of Pediatrics recommends no over-the-counter cough-andcold formulations for children younger than 6 years of age, because of lack of efficacy and potential for
harm.21,22
Conclusion
Use of specific techniques makes it easier for health-care providers to examine infants and children for common pediatric oral lesions. A careful medical history and strategic inspection of the mouth are essential for diagnosis of pediatric oral conditions. The most important considerations with pediatric oral lesions are treatment of pain and assessment of hydration. Consider referral
to an emergency department for dehydration, sepsis, or ill appearance, or when a PTA is a possibility.
Citation: Canares TL, Mudd SS. Pediatric oral lesions in the urgent care setting. Journal of Urgent Care Med. November 2016. Available at: https://www.jucm.com/pediatric-oral-lesions-urgent-care-setting/.
References
1. Centers for Disease Control and Prevention. Notes from the field: severe hand, foot and mouth disease associated with coxsackievirus A6—Alabama, Connecticut, California, and
Nevada, November 2011–February 2012. MMWR Morb Mortal Wkly Rep. 2012;61:213–214.
2. Ventarola D, Bordone L, Silverberg N. Update on hand-foot-and-mouth disease. Clin Dermatol. 2015;33:340–346.
3. Canares TL, Steele D. Oral lesions. In: Wolfson AB, Cloutier RL, Hendey GW, et al, eds. Harwood-Nuss’ Clinical Practice of Emergency Medicine. 6th edition. Philadelphia, PA: Lippincott
Williams & Wilkins; 2015: 1162–1163.
4. Arduino PG, Porter SR. Herpes simplex virus type 1 infection: overview on relevant clinico-pathological features. J Oral Pathol Med. 2008;37:107–121.
5. Hsiao CH, Yeung L, Yeh LK, et al. Pediatric herpes simplex virus keratitis. Cornea. 2009;28:249–253.
6. Amir J, Harel L, Smetana Z, Varsano I. Treatment of herpes simplex gingivostomatitis with aciclovir in children: a randomised double blind placebo controlled study. BMJ.
1997;314:1800–1803.
7. Nasser M, Fedorowicz Z, Khoshnevisan MH, Shahiri Tabarestani M. Acyclovir for treating primary herpetic gingivostomatitis. Cochrane Database Syst Rev. 2008(4):CD006700.
8. Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases
Society of America. Clin Infect Dis. 2012;55:1279–1282.
9. Le Marechal F, Martinot A, Duhamel A, Pruvost I, Dubos F. Streptococcal pharyngitis in children: a meta-analysis of clinical decision rules and their clinical variables. BMJ Open.
2013;3:e001482.
10. Wing A, Villa-Roel C, Yeh B, et al. Effectiveness of corticosteroid treatment in acute pharyngitis: a systematic review of the literature. Acad Emerg Med. 2010;17:476–483.
11. Waninger KN, Harcke HT. Determination of safe return to play for athletes recovering from infectious mononucleosis: a review of the literature. Clin J Sport Med. 2005;15:410–416.
12. Lennon P, Crotty M, Fenton JE. Infectious mononucleosis. BMJ. 2015;350:h1825.
13. Baldassari C, Shah RK. Pediatric peritonsillar abscess: an overview. Infect Disord Drug Targets. 2012;12:277–280.
14. Powell J, Wilson JA. An evidence-based review of peritonsillar abscess. Clin Otolaryngol. 2012;37:136–145.
15. Nystatin. Micromedex Solutions. Truven Health Analytics. 2016. Available from: https://www.micromedexsolutions.com
16. Pappas PG, Kauffman CA, Andes D, et al; Infectious Diseases Society of America. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious
Diseases Society of America. Clin Infect Dis. 2009;48:503–535.
17. Amir LH. Managing common breastfeeding problems in the community. BMJ. 2014;348:g2954.
18. Tharpe CA, Kemp SF. Pediatric allergic rhinitis. Immunol Allergy Clin North Am. 2015;35:185–198.
19. Pereira MS, Munerato MC. Oral manifestations of inflammatory bowel diseases: two case reports. Clin Med Res. 2016;14:46–52.
20. Feng S, Fan Y, Liang Z, et al. Concomitant corticosteroid nasal spray plus antihistamine (oral or local spray) for the symptomatic management of allergic rhinitis. Eur Arch Otorhinolaryngol.
2016;273:3477–3486.
21. American Academy of Pediatrics. Withdrawal of cold medicines: addressing parent concerns. 2008. Available from: http://www.aap.org
22. Smith SM, Schroeder K, Fahey T. Over-the-counter (OTC) medications for acute cough in children and adults in community settings. Cochrane Database Syst Rev. 2014;11:CD001831.
