Published on
Urgent message: With the increasing use of medications, especially antibiotics, more and more patients are presenting to urgent care with erythema multiforme. Correct diagnosis and identification of the underlying cause can result in rapid clinical resolution of the lesions.
Shailendra Kapoor, MD
Descriptions of erythema multiforme (EM) first appeared in the work of Albert and Bazin in 1822, but it was not until 1866 that von Hebra categorized these erythematous eruptions and labeled them “erythema exudativum multiforme.” Today, we know that EM is more common in younger adults, especially men.
There are two types of EM: EM minor and EM major. EM minor comprises nearly 70% of the cases. Most cases of EM minor resolve in one to three weeks, while EM major might take three to six weeks to resolve. Recurrences are more commonly seen in EM minor, but are rare in EM major. Traditionally, Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) were included in the same spectrum as EM.
However, the recent tendency has been to categorize SJS and TEN in a different category from EM. SJS and TEN usually involve the torso, and the Nikolsky’s sign is usually positive; in EM, the torso is usually spared and the Nikolsky’s sign is usually negative. The body surface area involved in TEN is greater than 30%, while in SJS and EM less than 10% of body area is involved.
Etiology and Pathogenesis
Even though the exact pathogenesis of EM is not completely understood, it is thought to be caused by viral, bacterial, or chemical triggers that initiate a hypersensitivity reaction. It may represent a type III immune complex-mediated hypersensitivity reaction, with a portion of the pathology arising from a type IV delayed hypersensitivity reaction. A majority of the patients with EM have deposits of complement 3, immunoglobulin M, and fibrin around the dermal blood vessels.
In early stages, a lymphocytic infiltrate is characteristically seen at the dermo-epidermal junction. The pathognomic finding in later stages is dermal edema along with lymphocytic infiltration (predominantly CD-4 cells) accompanied by epidermal necrosis (which may involve the entire epidermal thickness but is usu- ally predominant in the stratum basale). Satellite cell necrosis (i.e., lymphocytes surrounding necrotic keratinocytes) is another characteristic histological feature. Studies have shown that individuals with HLA-DQB1 are especially susceptible to the disease; HLA-B62, HLA- B35, and HLA-DQ3 are commonly seen in patients with recurrent EM.
Many different etiologies have been proposed in the pathogenesis of EM. Currently, herpes simplex virus (HSV) is thought to be the trigger in nearly 100% of cases of EM minor and nearly 50% of cases of EM major.3,4 Other viral causes include adenovirus, hepatitis, coxsackievirus, and echoviruses. Mycoplasma pneumo- niae infection is the most common bacterial trigger.5 Other bacterial causes include pneumococcus, Proteus, Neisseria, and Salmonella.
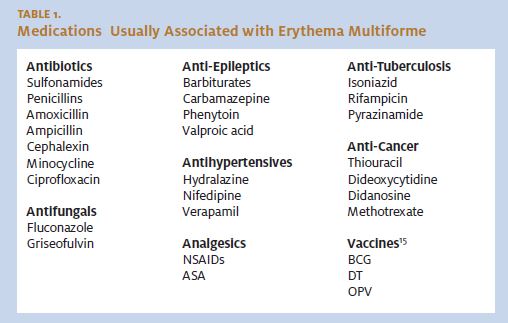
Drugs, especially sulfonamides, have been implicated in EM major. Some of the other drugs commonly implicated include NSAIDs, aspirin, barbiturates, phenytoin, and penicillin. (See Table 1.) Often, the etiology remains unknown.
Clinical Diagnosis
Symptoms
Most patients with EM minor present with new-onset mucocutaneous lesions which are usually symmetrical and rapidly progressing in nature. These lesions may be pruritic or may be associated with a burning sensation.
Skin involvement in EM major is usually preceded by prodromal symptoms such as fatigue, fever, headaches, and myalgias. These symptoms can appear up to two weeks prior to the mucocutaneous manifestations. Oral mucosal involvement may lead to difficulty in drinking and eating. Ocular involvement may lead to complaints of redness, discharge and ocular pain.
Signs
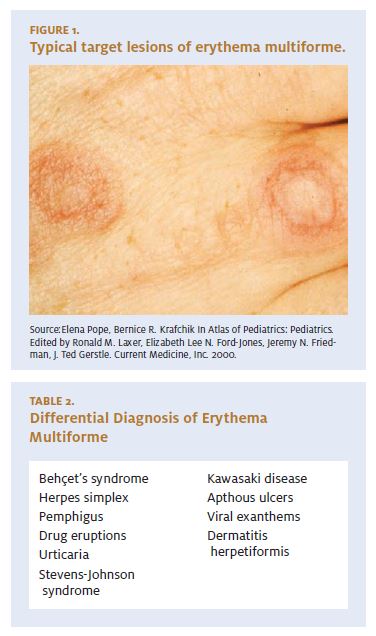
The initial skin lesion is an erythematous macule or papule, usually less than 3 cm. The hallmark of EM is a typical “target” or “iris” or “bull’s eye” lesion which consists of a dusky red center surrounded by an intermedi- ate pale and edematous ring6 (Figure 1). The periphery of the lesion gradually becomes violaceous giving rise to a concentric appearance. The greatest damage occurs at the center, with the peripheral rings showing lesser damage. Atypical “target” lesions consist of two rings in- stead of the usual three rings. These lesions are usually symmetrical and usually involve the palms, extensor surfaces of extremities, backs of hand, and feet, face, and neck. Involvement of the palms and soles is a characteristic feature of EM.
EM Minor
EM minor may be episodic or recurrent and is usually self limiting. Less than 10% of the body surface area (BSA) is involved in EM minor. Typically, Nikolsky’s sign is negative. Lesions last for one to three weeks and heal without scarring.
EM Major
Less than 10% of the body surface area (BSA) is involved in EM major. Nikolsky’s sign is negative. Lesions last for three to six weeks. The skin lesions are more severe, confluent, and vesiculbullous compared with EM minor. Mucosal lesions are seen in 40% to 60% of EM major cases. At least two mucosal surfaces must be involved to make a diagnosis of EM major. Mucosal involvement usually involves the lips and buccal mucosa and may present as bullae, ul- cerations with or without a pseudomembrane, or hy- perkeratotic plaques interspersed with erythematous changes.7 Ocular involvement may present as redness, discharge, swelling, corneal ulcers, anterior uveitis, and panophthalmitis. Usually, patients with EM major also have fever and generalized lymphadenopathy. Rarely, the genitourinary, gastrointestinal, and respiratory tracts may be involved.
Laboratory Tests
Usually, no laboratory tests are required for diagnosing EM minor. In EM major, elevated white blood cell counts, elevated erythrocyte sedimentation rate, and elevated acute phase reactants may occur. In severe cases, a basic metabolic panel and blood, skin, and mucosal cultures should be ordered to rule out renal involvement, electrolyte imbalances, and secondary infections. In patients in whom the diagnosis is uncertain, punch biopsy of the skin lesions should be performed. In early stages, a lymphocytic infiltrate is seen at the dermoepidermal junction; later stages are characterized by dermal lymphocytic infiltrates, epidermal necrosis, and satellite cell necrosis.
Complications
Most cases of EM minor resolve without any complications. Some of the complications that might occur include hyperpigmentation, hypopigmentation, or secondary bacterial infection. EM major is more likely to be associated with complications, especially in immunocompromised patients. Corneal ulcers, corneal opacities, anterior uveitis, panophthalmitis, conjunctival scarring, and blindness have been reported with ocular involvement. Severe systemic disease can lead to dehydration and electrolyte imbalances. Rarely, scarring may lead to stricture formation in bronchi, esophagus, urethra, and vagina. Besides the above-mentioned complications, myocarditis, nephritis, and respiratory failure can also occur rarely.
Differential Diagnosis
The differential diagnosis includes herpes simplex, which usually presents with predominantly vesicular le- sions and other dermatological conditions which might resemble EM, such as dermatitis herpetiformis, urticaria, drug eruptions, pemphigus, and Behçet’s syndrome (Table 2). Behçet’s syndrome manifests as recurrent aphthous ulcers, genital ulcerations, and uveitis.
Systemic diseases that may present with similar lesions include viral exanthems, septicemia, Kawasaki disease, and serum sickness. EM may also occur in patients with tuberculosis; in such a case, chest x-rays are helpful in establishing the diagnosis. Target lesions may also be seen in Lyme disease. However, the target lesions in Lyme disease are usually limited to the site of the tick bite.
In questionable cases, punch-skin biopsies are diagnostic.
Treatment
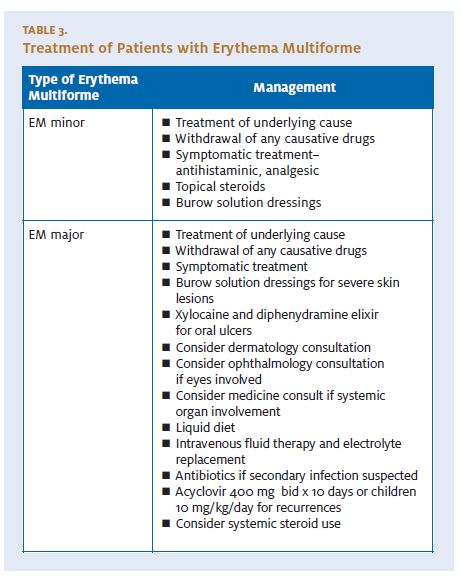
Treatment for EM minor and EM major is basically similar (Table 3). However, oral and ocular care may be an additional necessity if mucous membranes are involved in EM major. An emergency dermatologic consultation is indicated if it is unclear whether a patient has TEN, SJS, or EM. A dermatologic consultation, and possibly a subsequent skin biopsy, may also be necessary. The underlying cause, if identified, should be treated. If a medication is suspected, then it should be discontinued. Generally, mild cases are not treated. Symptomatic treatment involving oral antihist amines and analgesics is usually affective. Patients with mild symptoms are usually treated as outpatients.
Patients with severe cases should be admitted to a burn unit. Dehydration may also be severe. The clinician should be vigilant in monitoring electrolyte imbalances. Antibiotics may be necessary if secondary infection of lesions is suspected.
Skin Care
In mild cases, cold compresses and topical steroids can be used. Severe skin lesions should be treated as heat burns; 5% aluminum subacetate (Domeboro) solutions should be used and nonadherent dressings should be applied.
Oral Care
Viscous lidocaine or lidocaine gel can be used for pain re- lief in oral lesions. Diphenhydramine elixir may also be useful for oral lesions. Antibiotics may be necessary if secondary infections are suspected. A bland liquid diet may be necessary if eating and drinking are compromised by pain.
Systemic Steroids
Systemic corticosteroids may be considered in severe cases, though their use remains controversial. A one- to three-week course of prednisone is usually used. Prednisone (40 mg/day to 80 mg/day) is continued until control is achieved and is then tapered rapidly over a week.8,9 Treatment with prednisone may be successful in aborting a recurrence.
Antivirals
Acyclovir may be considered for prophylaxis for patients with more than five episodes per year. Doses are 400 mg twice a day, usually for six months. In children, a dose of 10 mg/kg/day is used. Herpes-associated EM is not prevented if oral acyclovir is administered after a herpes simplex recurrence is evident, and it is of no value after EM has occurred. Famciclovir and valacyclovir may be considered in patients resistant to acyclovir.
Alternative Treatments
If all the above treatments fail, thalidomide (100 mg/day), cyclosporine,10 immunoglobulins (0.75 g/kg/d for four days),11 azathioprine (100 to 150 mg/day), dapsone (100 to 150 mg/day),12 or interferon alpha13 can be tried.
Summary
Erythema multiforme is a hypersensitivity reaction usu- ally occurring one to two weeks after exposure to a drug or antigenic stimulus. Typically, it presents as a symmetrical, expanding, erythematous, maculopapular rash usually affecting the palms and the acral extensor surfaces. The typical lesion is described as the “target” or “iris” or “bull’s eye” lesion. The spectrum of the dis- ease ranges from EM minor, which is characterized by skin involvement usually sparing mucosa, to EM major, in which the lesions are larger and more confluent and the mucosa are usually involved. Most mild cases of erythema multiforme resolve without treatment; however, more severe cases may require hospital admission.14
References
- Rabelink NM, Brakman M, Maartense E, et al. Erythema multiforme vs. Stevens-Johnson syndrome and toxic epidermal necrolysis: An important diagnostic Ned Tijdschr Geneeskd. 2003;147:2089-2094.Roujeau Stevens-Johnson syndrome and toxic epidermal necrolysis are severity variants of the same disease which differs from erythema multiforme. J Dermatol. 1997;24:726-729.
- Aurelian L, Ono F, Burnett Herpes simplex virus (HSV)-associated erythema multiforme (HAEM): A viral disease with an autoimmune component. Derma- tol Online J. 2003;9:1.
- Weston Herpes-associated erythema multiforme. J Invest Dermatol. 2005;124:xv-xvi.
- Schalock PC, Brennick JB, Dinulos Mycoplasma pneumoniae infection as- sociated with bullous erythema multiforme. J Am Acad Dermatol. 2005;52:705-706.
- Wolf R, Matz H, Orion E, et al. Targeting the “target lesions.” Skinmed. 2005;4:311-312.
- Ayangco L, Rogers RS Oral manifestations of erythema multiforme. Der- matol Clin. 2003;21:195-205.
- Ferri Ferri’s Clinical Advisor: Instant Diagnosis and Treatment. 8thEd. St. Louis, MO: Mosby. 2006:309
- Yeung AK, Goldman Use of steroids for erythema multiforme in children. Can Fam Physician. 2005;51:1481-1483.
- Bakis S, Zagarella Intermittent oral cyclosporin for recurrent herpes sim- plex-associated erythema multiforme. Australas J Dermatol. 2005;46:18-20.
- Bachot N, Roujeau Intravenous immunoglobulins in the treatment of severe drug eruptions. Curr Opin Allergy Clin Immunol. 2003;3:269-274.
- Hoffman LD, Hoffman Dapsone in the treatment of persistent erythema multiforme. J Drugs Dermatol. 2006;5:375-376.
- Geraminejad P, Walling HW, Voigt MD, et Severe erythema multiforme responding to interferon alfa. J Am Acad Dermatol. 2006;54:S18-21.
- Huff JC, Weston WL, Tonnesen Erythema multiforme: A critical review of characteristics, diagnostic criteria, and causes. J Am Acad Dermatol. 1983;8:763-775.
- Frederiksen MS, Brenoe E, Trier Erythema multiforme minor following vaccination with paediatric vaccines. Scand J Infect Dis. 2004;36:154-155.

