Published on
Urgent message: While Boerhaave syndrome is a rare finding, a relatively high number of cases may present in the urgent care setting. As such, awareness of and vigilance for related symptoms are essential to taking a proper history and, ultimately, early diagnosis of acute, subacute, or chronic Boerhaave syndrome.
John Shufeldt, MD, MBA, JD, FACEP, Amber Hawkins, and Carli Nichta, MS4
Introduction
Boerhaave syndrome is a spontaneous esophageal rupture indicated in some cases by Mackler’s triad: subcutaneous emphysema, vomiting, and lower chest pain. Early diagnosis is critical for positive patient outcome, as this rare syndrome has a morbidity and mortality rate of 20%.1 With half the cases of esophageal ruptures being iatrogenic, the second most common cause being spontaneous perforation, history is an important part of diagnosis. Acute awareness of this rare syndrome and careful evaluation of patients with related signs and symptoms will help prevent missed diagnoses.
Case Presentation
A 41-year-old previously healthy female presents to the urgent care 12 hours after feeling like she had a piece of meat “stuck.” She states she initially gagged and then vomited x 1. She denies that the meat was expelled, however after the event she felt better. Subsequently, she developed midback pain that feels like a muscle strain and shoulder pain described as “achy.”
ROS is only positive for the back and shoulder pain. Past medical history is significant for hypertension, fibromyalgia, and obesity. She is status post lap-band procedure.
On her physical exam, her vitals are stable. She is afebrile. Other than her mild distress, her exam is normal. She has no abdominal tenderness, nor can you appreciate any abnormal heart or lung sounds. Chest x-ray is normal and the patient reports feeling better after a GI cocktail.
Introduction
The first historical record of spontaneous transmural esophageal rupture was documented by Dr. Herman Boerhaave regarding his patient Baron Jan Gerrit van Wassenaer.2 Following a large 3-day feast, Admiral van Wassenaer developed an upset stomach after a ride on his horse. He attempted to relieve his pain with an emetic, but while vomiting his pain suddenly increased exponentially and he reported that he felt as if something had ruptured. His pain would last 16 hours before he died of septicemia, and Dr. Boerhaave found the patient’s perforated esophagus on autopsy.
Boerhaave syndrome refers to a spontaneous transmural esophageal rupture from increased intra-esophageal pressure, specifically secondary to vomiting or straining.
Presentation
The classic presentation of Boerhaave syndrome is severe retrosternal chest and upper abdominal pain coupled with a history of significant retching or vomiting.3 These classic symptoms may be accompanied by a crunching, rasping sound occurring in synchrony with the heartbeat on auscultation, also known as Hamman’s sign. Hamman’s sign represents the presence of subcutaneous emphysema: when air from the ruptured esophagus infiltrates mediastinal tissues, the pressure of myocardial contraction creates a sound described by some as the “snap, crackle, pop” of Rice Krispies cereal. These three findings: evidence of subcutaneous emphysema, history of vomiting, and chest pain make up Mackler’s triad and are indicative of Boerhaave syndrome. Mackler’s triad, however, is only present in about half of the patients with esophageal perforation.4,5 Thus, despite the recognition of clinical findings specific to Boerhaave syndrome, the absence of Mackler’s triad is not evidence to rule out esophageal perforation.
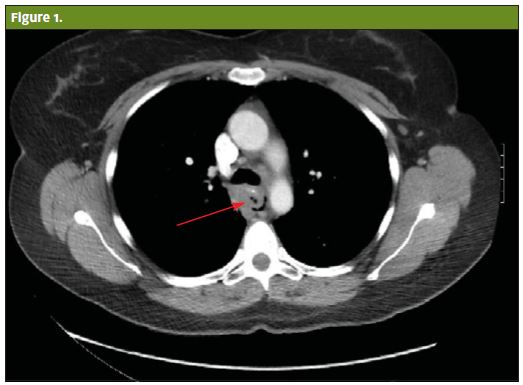
At initial evaluation, Boerhaave syndrome may be overshadowed by more prevalent options on the differential diagnosis. Chest pain at initial presentation is always concerning for a myocardial infarction and will often warrant a thorough cardiac workup. It is important, however, to maintain a high index of suspicion for esophageal perforation, especially when the chest pain localizes retrosternally with radiation to the back or left shoulder, or in the presence of a negative cardiac workup. One case report describes an esophageal perforation with complicated mediastinal involvement that presented with ECG changes of ST elevation in leads I, II, and a VL.6 Another potential convoluting factor is the association of nausea and vomiting with both myocardial infarction and esophageal perforation. Therefore, it is important to focus on obtaining a detailed, chronological history of symptom presentation, and assessment of risk factors.
A common misconception regarding life-threatening conditions is their presentation as acute, obvious, and easily identifiable. Many cases of Boerhaave syndrome will present in an urgent care environment. Furthermore, Boerhaave syndrome cases may present as acute, subacute, or chronic.
- Acute Boerhaave syndrome is defined as the development and presentation with symptoms within 24 hours of esophageal rupture.
- Subacute presentation occurs between 24 hours and 2 weeks following esophageal rupture.
- Chronic Boerhaave syndrome presents with subtle development of symptoms weeks to months after rupture.
Diagnosis of subacute and chronic Boerhaave syndrome is further complicated by the challenge of ascertaining an accurate history. To the typical patient there stands a significant disjunction between vomiting and the development of chest pain. Additionally, the more time passes between an initial vomiting event and the onset of symptoms secondary to esophageal perforation, the less related the two seem—certainly to the patient, and potentially to the evaluating provider.
Patients presenting with subacute and chronic cases of Boerhaave syndrome, as well as elderly patients, have both been shown to be less likely to report a history of vomiting at diagnosis of Boerhaave syndrome. In the face of the nonspecific symptoms and elusive diagnostic findings, often history plays a key role in diagnosing esophageal perforation. Early damage occurs within the first 6 hours, with the most damage being at around 12 hours.7
Other presentations include pleural effusion (not uncommon presentation), duodenal ulcer perforation, tension pneumothorax, hydrothorax, pneumomediastinum, and collapse of the lung.
Risk Factors
Risk factors for Boerhaave syndrome are intimately linked to its pathogenesis of severe vomiting. Vomiting severe enough to cause esophageal perforation is most often seen with alcohol abuse and bulimia, though any event of sustained or single emesis has the potential to precipitate Boerhaave syndrome. In addition, men between the ages of 50 and 70 years are the most likely candidates for esophageal perforation. Often, the patients present with sepsis.7
Interestingly, the most common cause of esophageal perforation is iatrogenic, generally implicated during intubation procedures; however, 6.8% of esophageal perforations are spontaneous, and can be life-threatening if unrecognized and untreated.8 Other known cases are due to weight lifting, labor, epileptic episodes, straining for bowel movements, use of the Heimlich maneuver, external trauma, perforating trauma, and ingestion of caustic substances.1,9
A related condition, Mallory Weiss syndrome, is also an esophageal pathology. Mallory Weiss syndrome, however is a longitudinal esophageal tear that does not extend beyond the mucosa, whereas Boerhaave is a full thickness perforation. Mallory Weiss syndrome is always associated with hematemesis, a strong distinction from Boerhaave syndrome, which rarely has associated hematemesis.10
Evaluation
Evaluation of esophageal perforation begins with a chest x-ray. Despite its availability and popularity, plain film radiographs are unreliable for diagnosis or rule-out of Boerhaave syndrome. When present on chest x-ray, pneumomediastinum, or free air in the mediastinum, gives a fairly limited differential diagnosis of esophageal rupture, asthma or alveolar rupture, bowel perforation, or Mycoplasma pneumoniae pneumonia. A detailed history would identify the diagnosis of Boerhaave syndrome in the presence of pneumomediastinum. A leak can sometimes be identified if the chest x-ray can include a water-soluble contrast solution instead of Barium, due to additional inflammation risk. In some cases, patients are unable to swallow, so a CT scan can also be used to reveal an esophageal perforation and potentially provide more information about its location. However, one study showed a false-negative rate of 15% to 25% using a CT scan or esophagogram with water-soluble contrast.10
Recently, the Pittsburgh group researched and published a decision-making protocol for evaluating esophageal perforations: the perforation severity score (PSS). Based on preexisting esophageal pathology and clinical presentation, three groups were determined: low-, intermediate-, and high-severity perforation. The PSS was developed to guide decision-making in areas with different morbidities and outcome strata. The PSS evaluates age, tachycardia, leukocytosis, pleural effusion, noncontained leak, respiratory compromise, and time to diagnosis >24 hours and guides decision-making mainly with regard to proceeding with operative vs conservative management. While both useful and successful, the PSS assumes a known diagnosis of esophageal perforations.
Treatment
Early diagnosis and proper care are critical for survival of patients with Boerhaave syndrome. In some cases, nonsurgical treatment is best. Patients should be transferred to the ICU and/or surgery for further evaluation and treatment. Nonsurgical treatment involves the patient being restricted to NPO and given IV fluid, antibiotics, protein pump inhibitors, and pain treatment; in some cases, a T-tube oesophagostomy is appropriate for reinforcement and to allow the damaged tissue to heal.1
After the patient has stabilized, another treatment option, depending on severity, is surgical repair and close follow-up. Spontaneous rupture leads to a complicated and challenging surgery. Surgical treatment can be risky and, in the case of spontaneous perforation, the mortality rate is high and a function of length of time since onset. Surgery within <24 hours has a mortality rate of 36%; >24 hours, this number increases to 64% according to one study.11 In all cases, the sooner the diagnosis and treatment onset, the better the patient outcome.
Case Resolution
In the present case, the chest x-ray performed in the urgent care was negative for mediastinal air. Because the provider had a high index of suspicion, the patient was transferred to the ED. At the hospital, the esophagogram was negative for both mediastinal air or extravasation. The CT of her chest, however, was positive for air in her mediastinum. She was admitted to the ICU after undergoing operative repair. Because of the early diagnosis, the patient made an uneventful recovery.
Citation: Shufeldt J, Hawkins A, Nichta C. Boerhaave Syndrome in a 41-year-old female. J Urgent Care Med. December 2017. Available at: https://www.jucm.com/boerhaave-syndrome-41-year-old-female/.
References
- Søreide JA, Viste A. Esophageal perforation: diagnostic work-up and clinical decision-making in the first 24 hours. Scand J Trauma Resusc Emerg Med. 2011;19:66.
- Adams BD, Sebastian BM, Carter J. Honoring the admiral: Boerhaave-van Wassenaer’s syndrome. Dis Esophagus. 2006;19(3)146-151.
- Salo J, Sihvo E, Kauppi J, Räsänen J. Boerhaave’s syndrome: lessons learned from 83 cases over three decades. Scand J Surg. 2013;102(4):271-273.
- Wei L, Wang F, Chen S. A late diagnosed case of Spontaneous esophageal perforation in an elderly patient. Int J Clin Exp Med. 2015;8(7):11594-11597.
- Dżeljilji A, Rokicki W, Rokicki M. A rare case of duodenal ulcer perforation accompanied by Boerhaave syndrome. Polish J Cardio-Thoracic Surg. 2015;12(3):262-265.
- Shemesh A, Taub CC. Inferolateral ST-segment elevation in Boerhaave syndrome. Am J Med. 2016;129(3):e27-e28.
- Kaori Kimura-Kataoka K, Fujihara J, Kurata S, et al. An autopsy case of spontaneous esophageal perforation (Boerhaave syndrome). Leg Med (Tokyo). 2016;23:5-9.
- Lindenmann J, Matzi V, Neuboeck N, et al. Management of esophageal perforation in 120 consecutive patients: clinical impact of a structured treatment algorithm. J Gastrointest Surg. 2013;17(6):1036-1043.
- Kaman L, Iqbal J, Kundil B, Kochhar R. Management of esophageal perforation in adults. Gastroenterol Res.2010;6(3):235-244.
- Vallabhajosyula S, Pranathi R. Sundaragiri PR, Berim IG. Boerhaave syndrome presenting as tension pneumothorax: first reported North American case. J Intensiv Care Med. 2016;31(5):349-352.
- Rokicki M, Rokicki W, Rydel M. Boerhaave’s syndrome—over 290 yrs of surgical experiences. Pol Przegl Chir. 2016;88(6):365-372.



