Published on
Urgent message: Care can be delayed if an urgent care or emergency clinician attempts to interrogate the CIED of a patient who does not know their device manufacturer and does not carry their ID card. This scenario illustrates the importance of patient education in care centers, such as the emergency department and urgent care.
Tinh M. Le; James F. Neuenschwander, MD, FACEP; Mary Jones, DNP; Ankur Parekh; Hana Le; Kaitlyn Cedoz; and Clark Daugherty
ABSTRACT
Background
Pacemakers and implanted cardioverter-defibrillators (also known as cardiac implanted electronic devices, or CIEDs) provide lifesaving functions and record critical clinical data. Clinicians cannot access this data or assess functionality without knowing the device’s manufacturer. Every CIED patient is given an identification card indicating the manufacturer. Patients presenting to emergency departments/urgent care centers (ED/UC) without ID cards can cause delays, requiring time to be spent contacting manufacturers. To our knowledge, no studies have examined the rate at which patients present to ED/UCs with their identification cards. This study’s purpose was to determine the rate at which CIED patients presented to an ED/UC with their ID cards.
Methods
The study site was a community hospital with an annual ED/UC census of over 70,000 patients. After obtaining IRB approval, a convenience sample was used to find participants. Patients that met inclusion criteria were surveyed.
Results
One hundred and six patients met inclusion criteria and were enrolled from June 2013 to September 2014. Fifty-eight percent were male. Male mean age was 72 (SD = 13.70), with a range of 40-95. Women had a mean age of 74 (SD = 16.92), (95% CI, 69.79- 75.55), and had a broader age range of 24-91. Overall, 58 patients (55%) presented with their ID cards. Twelve patients (11%) presented with a potentially device-related complaint. Of those 12, eight presented with their ID cards. Statistical analyses were performed to determine whether the age of individuals, the sex of the individuals, and the reason for presenting to the ED/UC made a significant difference between the rates at which ID cards were presented.
Conclusion
Fifty-five percent of CIED patients presented to the ED/UC with their device ID cards. Even in the group of patients with potentially device related complaints, only 66% presented with their respective ID cards.
INTRODUCTION
Almost 2 million patients in the United States live with cardiac implanted electronic devices (CIEDs), a term used to describe pacemakers and implantable cardiac defibrillators.1 CIEDs are indicated to treat a variety of cardiac arrhythmias. Pacemakers maintain a patient’s heart rate to ensure effective circulation, while implanted cardiac defibrillators provide voltage shocks to terminate life-threatening arrhythmias.2 As of 2016, roughly 200,000 pacemakers were implanted annually in bradycardic patients in the U.S.3 Worldwide, it is estimated that 1.25 million pacemakers are implanted annually.4 Because of their lifesaving functions and widespread use, it is essential for clinicians to be able to interrogate CIEDs. CIED interrogation reports include CIED data, such as recent arrhythmias or shocks, and allow providers to assess CIED functionality, such as device settings and battery life.
There are two classes of device used to interrogate CIEDs: device programmers, which can only be safely operated by International Board of Heart Rhythm Examiner (IBHRE)-trained technicians (often company representatives), and read-only device interrogators, which can be safely used by any healthcare provider. Regardless of the chosen interrogation method, healthcare providers must first have knowledge of the device’s manufacturer. This is because each of the three major CIED manufacturers (Abbott Laboratories., Boston Scientific Corporation, and Medtronic plc.) produce programmers and read-only interrogators that are only capable of interrogating CIEDs produced by that company. Consequently, each CIED patient is given an identification card (ID card) which indicates the device’s manufacturer.
Care can be delayed if an urgent care or emergency clinician attempts to interrogate the CIED of a patient who does not know their device manufacturer and does not carry their ID card. Often, all three possible manufacturers must be contacted; this is a time-consuming process.5 Bayley, et al in 2005 reported that delays in patient care can cause overcrowding in the emergency department and urgent care centers and interfere with potential need for admitting a patient into an inpatient bed.5,6 Because of this, it is crucial for CIED patients to carry their ID cards at all times.
To our knowledge, no study has examined the rate at which CIED patients present to the ED/UC with their ID cards. The purpose of this study was to determine the rate at which CIED patients present to the emergency department/urgent care (ED/UC) with their ID cards, and to test for differences between those that presented with and without their cards.
METHODS
An observational study was conducted to determine the rate at which CIED patients presented to the ED/UC with their device ID card. This study took place at a community hospital located in the Midwest with an annual ED/UC census of roughly 70,000 patients. The study was approved by the Institutional Review Board (IRB), was nonfunded and investigator-initiated, and was completed in accordance with STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) guidelines.7
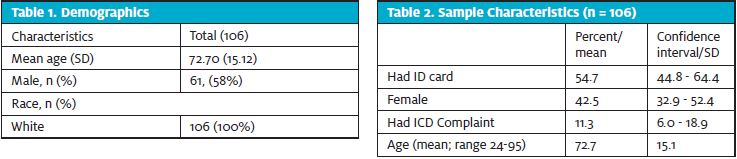
When trained research staff were available, all CIED patients presenting to the ED/UC were assessed for study inclusion. Inclusion criteria included: patients who had a CIED in place, who were able to answer whether or not they had their ID card, and who were over the age of 18. Prisoners, pregnant women, non-English speaking patients and those unable to respond to questions were excluded from the study. One hundred and six patients met the inclusion criteria and were enrolled from June 2013 to September 2014. Research staff determined whether or not patients had presented with their ID cards and recorded patient demographic information and chief complaints on a standardized data collection form (see Table 1).
Statistical analysis was performed using t-test for continuous variables and chi-squared test for categorical variables. We tested for significant differences between the ages of patients who presented with and without their ID cards, the rates at which males and females presented with their ID cards, and the rate at which patients with and without cardiac-related chief complaints presented with their ID cards. Alpha was set to 0.05. No formal sample size analysis was performed.
RESULTS
Of the 106 patients who participated in the study, 55% presented to the ED/UC with their ID cards. The cohort was 58%, with an average age of 73 years (StDev = 15.1). See Table 2.
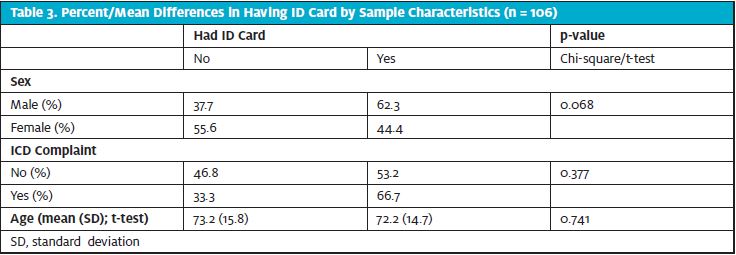
There was no significant difference between the rates with which male (62%) and female (44%) patients presented with their ID cards (p=0.068). See Table 3. Additionally, there was no significant difference between the rates at which patients with potentially device-related chief complaints, such as syncope or dyspnea, presented with their ID cards compared to patients without device-related chief complaints (66% and 53%, respectively; p=0.38). Finally, there was no significant age difference between patients who presented with and without their ID cards (72.2 vs 73.2 years, p=0.74).
DISCUSSION
Emergency and urgent care clinicians frequently provide care to CIED patients with complaints such as chest pain, shortness of breath, fatigue, syncope, and dizziness, all of which could be caused by CIED malfunction (see Table 4). Although a variety of other conditions could also cause these symptoms, unidentified CIED malfunction can result in significant morbidity and mortality, making it important to rule out CIED malfunction as a potential cause. This is why it is crucial to interrogate CIEDs early in a patient’s stay.
Each CIED can only be interrogated by its specific manufacturer’s interrogator. For the 45% of CIED patients in our study that presented without their ID cards, immediate interrogation would be impossible. Hence, it is crucial to be able to find this information in other ways. Anecdotally, we find that it is often, but not always, possible to determine this information in a patient’s electronic medical record (ie, by reading provider notes from the patient’s electrophysiology appointments). However, if an out-of-town patient presents without their ID card and cannot remember their manufacturer, you can find this information by calling each manufacturer’s phone number and speaking with a representative:
- Abbott (formerly St. Jude): (1-800) 722-3774
- Boston Scientific: (1-800) CARDIAC (227-3422)
- Medtronic: (1-800) 929-404
The traditional methods listed above are effective, but often time-consuming, meaning that patients who present without their ID card can face significant delays in care.8 Recent studies have suggested an alternative method—the use of read-only CIED interrogators.Read-only interrogators are incapable of altering CIED function, and can be safely used by any care provider, obviating the need to call company representatives.9 Like CIED programmers, each CIED manufacturer also produces a read-only interrogator. If an ED/UC owns each of the major manufacturers’ read-only CIED interrogators, it is possible to determine an unknown CIED’s manufacturer by simply attempting to interrogate the device with each company’s interrogator. Only the correct interrogator will connect, bypassing the need for multiple phone calls and hold times.10
Patients presenting to ED/UCs may have complaints related to their CIEDs that require interrogation; therefore, the CIED manufacturer must be known. If patients carry their device ID cards, then their care may be expedited.
The results of this study are crucial to emergency and urgent care clinicians, as device ID card presentation can potentially allow for a more efficient interrogation process. Potential solutions to alleviate this issue include a phone app containing the necessary identification rather than a physical ID card. Additionally, the creation of a single call center for all three companies could expedite finding the device manufacturer and does not involve multiple attempts at contacting the manufacturer.
These results also demonstrate the importance of proper patient education. It is feasible that, with improved patient education both in the electrophysiology clinic and at ED/UC discharge, a given area’s CIED patient population could grow to better understand the importance of always carrying their ID cards.
LIMITATIONS
This study had several limitations. Firstly, no a priori power analysis was performed to determine ideal sample size. Secondly, because the study was conducted at a single community hospital system with a predominantly Caucasian patient population, the results of this study may be difficult to generalize. Similarly, the results may not be applicable to urgent care facilities that are not aligned with a healthcare system or those that do not have the capacity to interrogate a CEID. Finally, patients were sampled using convenience sampling due to the nature of research staff schedules.
CONCLUSION
About half (55%) of CIED patients presented to the ED/UC with their device ID cards. Even in the group of patients with potential device related complaints, only 66% of patients presented with their respective ID cards. No statistically significant difference was found relating to age, sex, or chief complaints. While several limitations impact the generalizability of our results, we identified a significant potential issue in the treatment of CIED patients.
REFERENCES
- Eyal A, Roguin A. Magnetic resonance imaging in patients with cardiac implantable
electronic devices. Circulation. 2015;132:e176-178. - Haghjoo, M. Cardiac implantable electronic devices. In: Maleki M, Alizadehasl A, Haghjoo M, eds. Practical Cardiology. 1st ed. St. Louis, MO: Elsevier; 2018:251-260.
- Van der Zee S, Doshi S. Permanent leadless cardiac pacing. American College of Cardiology, Expert Opinion. Available at: https://www.acc.org/latest-in-cardiology/articles/2016/03/23/08/09/permanent-leadless-cardiac-pacing. Accessed September 10, 2021.
- Carrión-Camacho MR, Marín-León I, Molina-Doñoro JM, González-López JR. Safety of permanent pacemaker implantation: a prospective study. J Clin Med. 2019;8(1):35.
- Neuenschwander JF, Peacock WF, Migeed M, et al. Safety and efficiency of emergency department interrogation of cardiac devices. Clin Exp Emerg Med. 2016;3:239-244.
- Bayley MD, Schwartz JS, Shofer FS, et al. The financial burden of emergency department congestion and hospital crowding for chest pain patients awaiting admission. Ann Emerg Med. 2005;45:110–117.
- Vandenbroucke JP, von Elm E, Altman DG, et al. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. Ann Intern Med. 2007;147:W163-194.
- Jarvis, Paul RE. Improving emergency department patient flow. Clin Exp Emerg Med. 2016;3(2):63-68.
- Neuenschwander JF, Peacock WF, Le TS, et al. 102 pacemaker and defibrillator interrogations in the emergency department and hospital rarely lead to device reprogramming. Ann Emerg Med. 2017;70:S41.
- Le T, Neuenschwander J, Sankoe M, et al. Interrogation of pacemaker/defibrillator with mismatched read-only interrogators does not cause malfunction. Academ Emerg Med. 2019;26(S1):S156.
Author affiliations: Tinh M. Le, Case Western Reserve School of Medicine. James F. Neuenschwander, MD, Genesis Healthcare System; The Ohio State University. Mary Jones, DNP, Genesis Healthcare System; Frontier Nursing University. Ankur Parekh, The Ohio State University. Hana Le, The Ohio State University. Kaitlyn Cedoz, Alabama College of Osteopathic Medicine. Clark Daugherty, University of Toledo College of Medicine and Life Sciences. The authors have no relevant financial relationships with any commercial interests. The principal investigator has a consulting agreement with Boston Scientific.
