Published on
Urgent message: Clinical suspicion of CA-MRSA should be high for any skin wound or soft tissue infection with delayed healing, abscess, or persistent cellulitis.
RALPH S. BOVARD, MD, MPH, and ANNE REINER, MD, MPH
Introduction
Caregivers need to remain vigilant and challenge diagnoses to avoid complacency in the treatment of “common” problems. We all need to foster the habit of systematic and meticulous clinical evaluation. As Goethe said: “We see what we know.”
Case Presentation
The patient was a 48-year-old male who presented to the Occupational Medicine (OM) clinic approximately 2 weeks following an injury to his posterior lower leg. While at work as a janitor for the local school district, he had leaned a folding table on its side against the wall. As he turned it fell, scraping and abrading the posterior aspect of his left lower leg for about 4 inches to just above the heel. He was able to walk to the workplace first aid station where it was treated as a superficial wound not requiring sutures.
Four days later, he noticed swelling of the posterior ankle and went to the local emergency room. X-rays were obtained which did not show evidence of a fracture (Figures 1 and 2). He was diagnosed with a superficial wound infection and started on cephalexin 500 mg po QID x 7 days. He was seen in our clinic 2 weeks post-injury for a return-to-work letter after having completed the cephalexin. The wound was reported as “improved” with less discomfort and swelling.
Pertinent History
The patient had no known allergies and did not take any medications on a regular basis. He was a pack-a-day smoker and had gained 30 to 40 lb in the past 3 years. He denied a history of type II diabetes and reported a normal fasting blood sugar test the previous year.
Follow-up Evaluation
Two weeks later (1 month after the initial injury) he returned to the OM clinic with a complaint of persistent swelling, redness, and drainage from the wound that had now increased in size. He was unable to get his foot into his work boot because of pain and swelling and thus was unable to work.
Physical Exam (1 month post-injury)
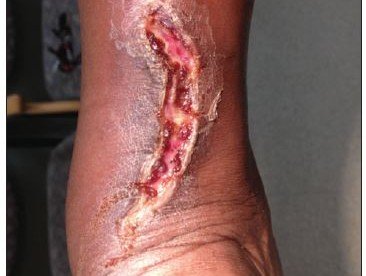
Height 6’2”. Weight 240 lb. On inspection there was a 7 cm x 1 cm curvilinear wound on the posterior left lower leg extending to just above the heel (Figure 3). The wound overlay the Achilles tendon, but the tendon was not visible beneath the wound. Thompson test was negative (normal). The wound showed some evidence of granulation but with yellowish, mucopurulent drainage. Range of motion at the ankle was 10 degrees DF and 45 degrees PF. There was some swelling extending to the lateral ankle but no focal tenderness over either malleoli. No signs of ligamentous injury.
Course and Treatment
Wound culture was performed and sent to the laboratory for analysis. We started the patient on Augmentin 875 mg po BID x 10 days and initiated twice-daily dressing changes with a non-adherent gauze, sterile bandages, and silver sulfadiazine 1% cream. He was to keep the wound clean and dry. He was placed on light duty work status.
Diagnostic Studies
On laboratory analysis the gram stain showed many gram-positive cocci and gram positive cocci in clusters. The wound culture showed many Staphylococcus aureus organisms resistant to Oxacillin.
WBC: 9.2k/uL HGB: 14.6g/dL HCT: 42.8% PLTS: 207k/uL.
Electrolytes: Normal except for fasting glucose 123 mg/dL.
GFR: Normal at >60 mL/min/1.73m2 CRP: <0.5 mg/dL
Cholesterol: 133 mg/dL; TRIG: 153 mg/dL (H), HDL 39 mg/dL (L).
Diagnosis
Community-Acquired (CA)-Methicillin Resistant Staphylococcus Aureus (MRSA) wound infection
Revised Treatment Plan
Our infectious disease service was consulted. The patient’s antibiotic coverage was changed, based on culture sensitivities, to sulfamethoxazole-trimethoprim 800 to 160 mg po BID x 14 days. He was instructed in proper wound care management and disposal of MRSA contaminated bandage material. He was referred to a nutritionist and to his primary care physician for type II diabetes management. He was seen for follow-up in the OM clinic at 1-week intervals over the ensuing month with improvement of symptoms and gradual healing of the ankle wound. He failed his final follow- up visit.
Discussion
When MRSA first emerged in the 1990s it was largely associated with hospitals and nursing homes. In recent years, it has emerged as an increasingly common cause of skin infections in otherwise healthy individuals. The proportion of staphylococcus infections attributable to MRSA is reported to have increased from 40% in 1999 to approximately 60% in 2005. MRSA is generally divided into community acquired (CA) and hospital acquired (HA) forms. CA-MRSA, first identified in 1998, is currently estimated to account for 14% of all MRSA cases reported. Groups at risk for CA-MRSA include members of the military, children in daycare, people who have gotten tattoos, athletes with frequent skin contact or who may share towels or razors, those who inject illegal drugs, and individuals with compromised immune systems (individuals with metabolic disorders such as type II diabetes mellitus may fall in this risk pool). Risk factors associated with HA-MRSA include recent hospitalization, surgery, residence in a long-term care facility, dialysis, or the presence of invasive medical devices.
The Agency for Healthcare Research and Quality (AHRQ) estimated that there were upwards of 390,000 cases of MRSA in the United States in 2005.1 Klevens, et al estimated 94,000 invasive MRSA infections requiring hospitalization and upwards of 19,000 deaths in 2005.2 The excess cost of treating a MRSA infection (versus a methicillin-sensitive S. aureus infection) ranges from $3,000 to $35,000 per case; this resulted in total costs to the health care system of $830 million to $9.7 billion in 2005.3 Patient education is important to limit the further spread of this disease to close contacts. Clean dry bandages, careful hygiene, proper disposal of dirty bandages, consistent use of gloves, hand washing, and alcohol or antiseptic gels are important modalities in limiting the further spread of MRSA.4 Nasal colonization has been identified as a potential risk factor for infection. Increasingly it is a pre-surgical consideration, especially in total joint procedures to test for nasal colonization. While 25% to 30% of individuals are colonized with staphylococcus, fewer than 2% are colonized with MRSA.5 Decolonization using topical and systemic antimicrobials and antiseptic body washes such as chlorahexadine are increasingly used as control interventions.
MRSA infections are classically resistant to beta-lactams (penicillins and cephalosporins), macrolides/azalides (erythromycin, clarithromycin, and azithromycin), and quinolones (levofloxacin). CA-MRSA has typically been responsive to gentamicin, tetracycline, doxycycline, clindamycin, and trimethoprim/sulfamethoxazole. HA-MRSA is often resistant to multiple classes of anti- microbial agents. Clinical suspicion of CA-MRSA should be high for any skin wound or soft tissue infection with delayed healing, abscesses, or a persistent cellulitis. CA MRSA may be confused with tissue necrosis associated with spider bites (rare). The severity of MRSA skin infections may vary from mild superficial infections to deeply invading soft tissue processes requiring hospital admission, surgical excision, and parenteral antibiotics. It is essential to obtain wound cultures in order to determine antibiotic sensitivities.6 Timely referral to infectious disease and specialty surgical services is indicated.
References
- 1 Elixhauser A, Steiner C. Infections with methicillin-resistant Staphylococcus aureus (MRSA) in U.S. hospitals, 1993-2005. HCUP Statistical Brief #35, June 2012. http://www.hcup- us.ahrq.gov/reports/statbriefs/sb35.pdf (accessed October 10, 2007).
- Klevens RM, Morrison MA, Nadle J, et al. Invasive methicillin-resistant Staphylooccus aureus infections in the United States. JAMA. 2007;298(15):1763-1771.
- Klein E D, Smith L, Laxminarayan Hospitalizations and deaths caused by methicillin- resistant Staphylococcus aureus, United States, 1999-2005. Emerg Infect Dis. 200713(12):1840-1846.
- Fishman N, Calfee Prevention and control of health care-associated infections. In: Goldman L, Schafer A, eds. Cedil Medicine. 24th ed. Philadelphia, PA: Saunders Elsevier; 2011; chap 290.
- Gorwitz RJ, Kruszon-Moran D, McAllister SK, et Changes in the prevalence of nasal colonization with Staphylococcus aureus in the United States, 2001–2004. J Infect Dis. 2008;197:1226-1234.
- C Liu, Bayer A, Cosgrove SE, et Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant Staphylococcus Aureus Infections in Adults and Children . Clin Infect Dis. 2011;52(3): e18-55.
