Published on
As lab testing for COVID-19 continues to evolve, new products—and new CPT codes—continue to emerge. The latest tests combine COVID-19 testing with a test for influenza A and B. There are two types: one includes the polymerase chain reaction (PCR) test, the other includes the antigen test. The new CPT codes are:
- 87636 Infectious agent detection by nucleic acid (DNA or RNA); severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (Coronavirus disease [COVID-19]) and influenza virus types A and B, multiplex amplified probe technique
- This CPT is effective as of October 6, 2020. The CPT Assistant article can be found at https://www.ama-assn.org/system/files/2020-10/cpt-assistant-guide-coronavirus-october-2020.pdf.
- 87428 Infectious agent antigen detection by immunoassay technique, (eg, enzyme immunoassay [EIA], enzyme-linked immunosorbent assay [ELISA], fluorescence immunoassay [FIA], immunochemiluminometric assay [IMCA]) qualitative or semiquantitave; severe acute respiratory syndrome coronavirus (eg, SARS-CoV, SARS-CoV-2 [COVID-19]) and influenza virus types A and B
- CPT 87428 is effective as of November 10, 2020. This CPT Assistant article can be found at https://www.ama-assn.org/system/files/2020-11/cpt-assistant-guide-coronavirus-november-2020.pdf.
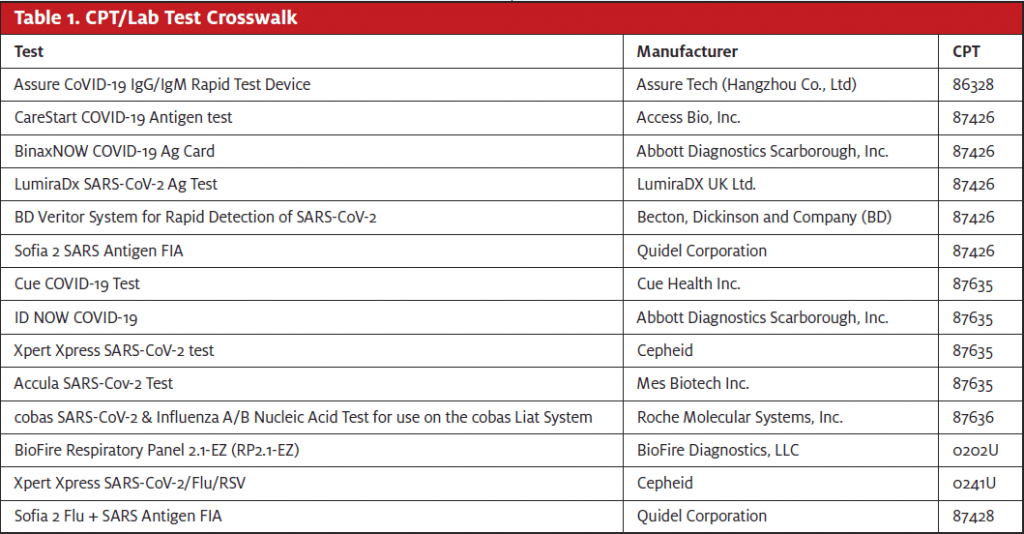
The new cobas SARS-CoV-2 & Influenza A/B Nucleic Acid Test for use on the cobas Liat System that is manufactured by Roche Molecular Systems, Inc. is reported with CPT 87636. Quidel Corporation has a test, Sofia 2 Flu + SARS Antigen FIA, that is reported with CPT 87428. See Table 1 for a CPT/lab test crosswalk. All the tests listed have a waived status. Modifier QW should be appended when the insurance plan requires it.
While these new CPT codes are needed in a timely fashion, they present unique problems for payers and the RCM team. These are payable codes by insurance plans, but payers are looking to Medicare to set a fee at the national level. Instead, CMS has made them contractor-priced until they undergo the CLFS annual payment determination process.
Take code 87426 for antigen testing as an example. Here is a summary of what we’re seeing at Experity for reimbursement:
- Overall average reimbursement is in the high $30s or low $40s.
- Several BCBS plans (eg, Oklahoma, Indiana, Kentucky) are driving the average down from the median allowable of $45.23 due to allowables between $9 and $12 using a flawed pricing formula. This may change if/when we do get a national price.
- The cost for most clients is in the low-to-mid $20s, so profit is going to be driven by payer mix considering the variability in payer pricing.
- The HRSA Uninsured Portal is holding claims with CPT 87426 until a national allowable is set by CMS. It will be covered though payment is delayed.
Due to this confusion, practices are wondering if they can just have patients pay for the test instead of billing their insurance. The short answer is no. Providers under contractual agreement with a payer to provide covered services are contractually obligated to bill insurance for those services and to only collect allowable patient portions based on the individual member benefit. Example:

Additionally, payers paying off case rates/global rates/flat rates consider COVID testing to be not billable outside of the flat rate. If COVID testing is done in the office as part of the office visit, the payer considers the case/global/flat rate as payment in full. Payers consider it a violation of the agreement if a provider charges the patient separately in this case. However, sending the patient out for testing is acceptable.
Experity is actively monitoring this situation.
Vaccines
In anticipation of a vaccine for COVID-19, the American Medical Association has worked with the Food and Drug Administration, Centers for Disease Control and Prevention, and the Centers for Medicare & Medicaid Services to create a new code set for these services. The plan is to use these for both billing and tracking. They can be found in a new Appendix Q, which will be updated as new vaccines become available (https://www.ama-assn.org/system/files/2020-11/covid-19-immunizations-appendix-q-table.pdf).
The structure is different from other vaccines. Each unique vaccine will have its own administration codes(s), depending on the number of doses. The manufacturer and name will be specifically listed in CPT, along with the NDC.
For example, Pfizer’s vaccine will be reported as follows:
First dose:
- Vaccine: 91300
- Admin: 0001A
- NDC: 59267-1000-01
- ICD: Z23
Second dose:
- Vaccine: 91300
- Admin: 0002A
- NDC: 59267-1000-01
- ICD: Z23
Those administration codes can only be used with CPT 91300 for the Pfizer product.
This Appendix Q will expand as new vaccines become available. If you change products, you need to change codes.
There is a lot we don’t know from an RCM perspective on what clinics will bill and to whom. Experity continues to monitor and will provide information as it becomes available.
Looking forward to a healthy and social 2021!

