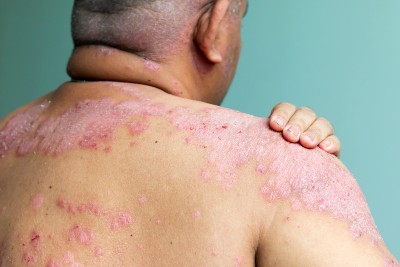
The Food and Drug Administration (FDA) has approved the oral medication icotrokinra for the treatment of moderate-to-severe plaque psoriasis in adults and pediatric patients 12 years of age and older, according to a press release. An interleukin-23 receptor antagonist, the once‑daily medication is a uniquely targeted oral peptide for patients who weigh at least 40 kg and who are candidates for systemic therapy or phototherapy. New guidance from the International Psoriasis Council clarifies when clinicians …
Read More