Published on
Urgent message: The last 10 years have seen a dramatic increase in DNA parentage testing. Yet, the DNA testing industry remains, in essence, unregulated. Can urgent care providers fulfill a need for ethical and clinically reliable access, while receiving direct and immediate payment for your services?
Elizabeth Panke, MD, PhD
For many generations, defining the parent-child relationship was easy. The woman who gave birth to the child was the child’s legal mother; to a large extent, fatherhood was assigned on a social basis.
With the advent of DNA testing, however, the social definition of fatherhood is increasingly being replaced by a genetic test. Advances in the technology employed in DNA testing have pushed the issue of fatherhood into the public spotlight—witness the tabloid-fed frenzy surrounding the paternity of the late Anna Nicole Smith’s child—and forced many to re-evaluate the definition of “family.”
The American Association of Blood Banks reports that more than 1 million persons undergo DNA parentage testing each year, with double-digit growth each year. The growth of commercial DNA-based parentage testing has been further spurred by our social and legal systems.
The federal Child Support Enforcement and Paternity Establishment Program was created in 1975 to help establish paternity for a growing number of non-marital children and to support collection of child support payments. The Family Support Act of 1988 requires states to have all parties in a contested paternity case take a genetic test upon the request of any concerned party.
More recently, the states of Ohio and Georgia, among others, have passed legislation that relieves a man of all parental obligations if a DNA test can prove that he is not the child’s biological father, regardless of the age of the child or the number of years the man previously was acknowledged and considered the child’s father. A growing list of U.S. court systems allowing such disestablishment of paternity is tracked by the National Conference of State Legislatures and can be viewed on their web site.
.4 Recent years have also seen the creation of the fathers’ rights groups.5 These groups have garnered attention on the Internet and on television shows hosted by the likes of Sally Jessy Raphael, Montel Williams, Jenny Jones, Maury Povich, Ricki Lake, and others. Marketing efforts by some DNA testing laboratories also have contributed to the reinforcement of the belief that DNA testing is a natural method for determination of the essence of identity.
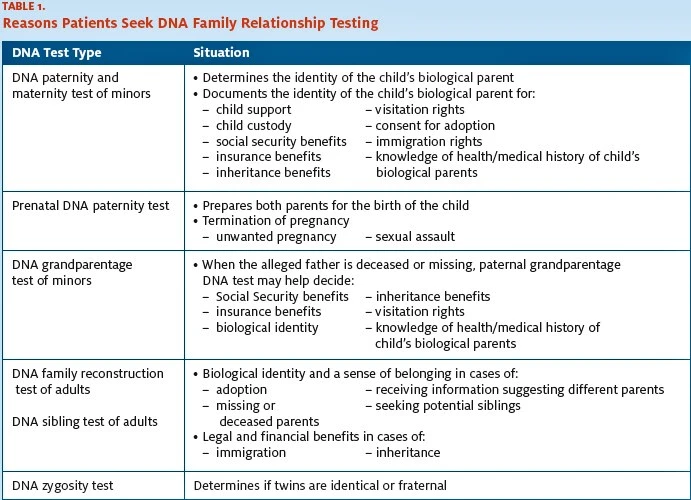
Reasons Patients Seek DNA Paternity or Family Relationship Testing
Patients may seek genetic family relationship testing for any number of reasons (Table 1). However, highly accurate and reliable prenatal DNA paternity tests may also be conducted using fetal cells from amniotic fluid (5 ml to 10 ml) or chorionic villus sampling (CVS) if a woman is undergoing amniocentesis or CVS for medical reasons. Patients who have no medical indication for those procedures should receive information on the risks associated with amniocentesis and CVS and should be given the option of having paternity testing per- formed after birth, which is just as accurate as pre- natal testing.
The DNA paternity testing laboratory can utilize the cultured fetal cells from the chromosomal lab to con- duct the testing. Typically, samples from the mother and from the alleged father are collected with a buccal swab.
The Role of Healthcare Providers
A national consensus for the choice providers of DNA testing services to the public has not yet been established. Federal regulation of laboratories under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) does not extend to DNA family relation- ship testing, since such testing is not considered medical testing.6
In contrast, the New York State Department of Health mandates that all DNA specimen collections and DNA family relationship tests on New York residents must be authorized by a licensed physician, and that DNA parentage test results must be released only to the ordering physician.7
This lack of consensus leaves most patients unable to identify reliable providers of these services. When seeking DNA testing services, some patients turn to physicians or family law attorneys for a referral to a DNA testing laboratory. Other patients consult their yellow pages and/or the worldwide web to identify providers of DNA parentage testing services.
Contrary to the common—and naïve—public trust, DNA family relationship testing is largely an unregulated industry. Government regulation of parentage testing is limited, and DNA paternity laboratory inspections and accreditation are voluntary. There is no mandatory over- sight of DNA parentage testing facilities.
In fact, many businesses market DNA paternity testing services and collect specimens, but outsource the actual testing to various laboratories (often, simply the lowest bidder), unbeknownst to the patients.
Recently, the Food and Drug Administration, Federal Trade Commission, and the Centers for Disease Control and Prevention developed a consumer alert that encourages consumers to consult their healthcare provider be- fore using direct-to-consumer genetic tests.8
It is reasonable to suggest that patients would be well served by having their initial contact for the delivery of DNA testing services be with their physicians and healthcare providers. Given the breadth of services they provide, it is reasonable to suggest that urgent care clinicians may be well suited to fulfill such a need.
A Partnership Model
How might such a partnership between the clinician and DNA testing laboratory work? It could be reasoned that healthcare providers should serve as the patients’ first contact for the provision of these services and be able to help patients decide whether DNA family relationship testing is needed, the type of genetic test that may best be suited to their situation, and the selection of a competent DNA testing facility. The healthcare provider could then collect DNA specimens and ship the specimens to the DNA testing lab- oratory for processing. The DNA testing laboratory per- forms the testing, releases the results per instructions of the healthcare provider, and provides 24-hour physician consultations on the interpretation of the results to the healthcare provider and/or the patient. The clinician may also be able to help patients understand the test results and offer guidance on the availability of qualified counseling in cases where such counseling is needed. In such a model, the patients pay healthcare providers directly for the clinical consultations and DNA specimen collections at the time they initiate these services. The DNA testing facility receives payment for the laboratory service they perform. The average national cost to the patient for legally admissible DNA paternity test results ranges from $350 to $500.
Thus, delivery of DNA family relationship services in a partnership between a healthcare provider and a reliable DNA testing laboratory is a win-win situation for the patient, the healthcare provider, and the laboratory: The patient receives competent, professional clinical care, reliable DNA test results, and a referral to professional counseling assistance, if needed; the testing lab- oratory gains patient specimens while knowing that their services are delivered to patients with comprehensive care before, during, and after the DNA test; and the healthcare provider has the satisfaction of providing much needed patient care and receives immediate, direct payment for services rendered.
Selecting a Partner Laboratory
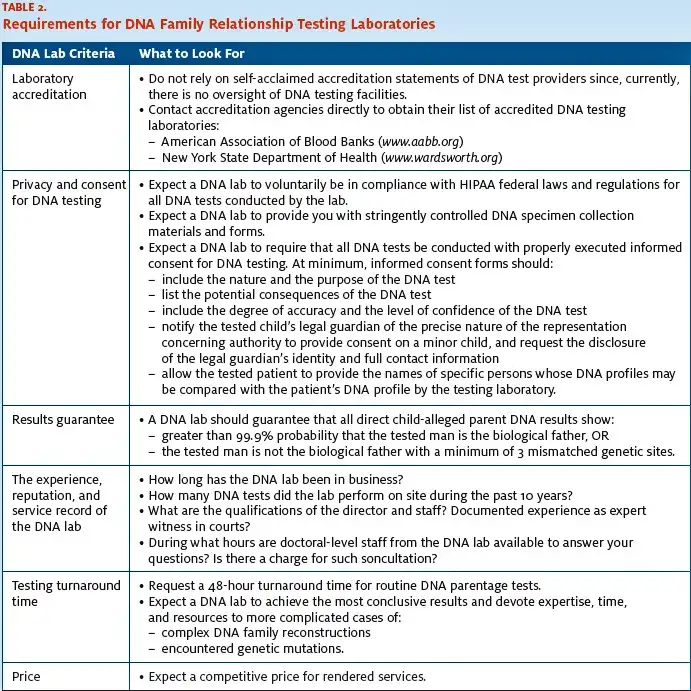
Because there is no regulatory oversight of DNA parent- age testing facilities, healthcare providers can be especially helpful in guiding patients in their selection of a qualified DNA testing laboratory. Such DNA testing facility should, at minimum, meet the requirements described in this section and summarized in Table 2.
Laboratory Accreditation
A seal of accreditation informs you and your patients that the DNA testing facility, at minimum, meets the established, national testing standards in conducting a DNA test. The most reliable way to determine the accreditation status of a DNA testing laboratory is to contact the national accreditation agencies directly and obtain their list of accredited DNA testing laboratories. Examples of such accreditation agencies include the following:
- The American Association of Blood Banks (AABB) offers a comprehensive list of parentage testing facilities it has accredited at aabb.org; from the association’s homepage, click on Accreditation, then on Parentage Testing Accreditation Program, and finally on AABB Accredited Parentage Testing Laboratories. Alternatively, call the AABB at (301) 215- 6584 to determine if a DNA laboratory has been accredited by the AABB.
- If your patients reside in New York, the DNA testing laboratory must be accredited by the New York State Department of Health (NYSDOH). You can determine if the DNA testing laboratory has such accreditation by going to wadsworth.org and clicking on Quality Certification, then on Clinical Laboratory Evaluation Program, and Approved Laboratories. Last, click on Parentage/Identity Testing – DNA Testing.
Privacy, Security of Information, and Informed Consent
Federal regulation of laboratories under CLIA does not extend to parentage testing laboratories,6 and there is a wide variation in the manner with which DNA testing laboratories handle areas such as privacy, confidential- ity, and informed consent.9
Healthcare providers who enter into business partner- ships with DNA testing laboratories need to know if their associate DNA testing laboratory routinely con- ducts DNA testing on specimens collected from patients without documented consent for such testing. Clearly, it is in the provider’s best interest to choose a DNA testing laboratory that has adopted policies that are in compliance with the Health Insurance Portability and Accountability Act (HIPAA) and regulations for all DNA tests performed by the laboratory. This means providing healthcare providers with stringently controlled DNA specimen collection materials and requiring that all DNA testing be conducted with properly executed informed consent.
The informed consent forms provided by the DNA testing laboratory for family relationship testing should, at minimum, describe the nature and the purpose of the DNA test, the potential consequences of the test, and the degree of accuracy and the level of confidence that can be expected. The informed consent form should also allow the tested patient to provide the names of the specific individuals whose DNA profiles may be compared with the patient’s DNA profile by the testing laboratory.
In cases where minor children are tested, the form should notify the child’s legal guardian of the precise nature of the authority to provide consent for DNA testing on a minor child, and should request disclosure of the legal guardian’s identity and full contact information.
Results Guarantee
The general public may have the impression that DNA testing is infallible. However, erroneous DNA test results are not uncommon.
False negative results most commonly arise from human error committed during specimen collection or during the DNA testing process.
False positive results often stem from the limited amount of DNA testing that is conducted by the laboratory. If a DNA testing laboratory tests a limited number of genetic sites in a DNA paternity test, the tested child and man may match many individuals within the population on every tested gene. Such DNA testing may falsely substantiate fatherhood.
The accuracy of DNA testing increases by testing in- creasing numbers of genetic sites. Some scientists—including the author—may believe that AABB and NYS- DOH standards requiring the accredited laboratories to reach a probability of paternity of only 99% during DNA testing is set too low.9
To produce the most conclusive DNA results, request that the laboratory not stop testing until they produce one of the following results:
- a probability of greater than 9% that the tested man is the biological father of the child; or
- a minimum of three mismatched genetic sites that prove that the tested man is not the biological father of the
Request that the laboratory guarantee that you will receive only one of these DNA results.
Experience, Reputation, and Service Record
Lab Personnel
Important issues to consider when selecting a DNA testing laboratory also include the experience and qualifications of the laboratory director, staff, and technicians performing the testing. A laboratory that has been performing DNA parentage testing for many years is more likely to have experienced staff and well-validated testing procedures; such a laboratory is also more likely to stay in business for the foreseeable future.
Ask how long the laboratory has been in business, how many DNA paternity tests the laboratory has performed on site during the past 10 years, the qualifications of the director and staff, and how much experience they have in providing expert witness testimonies in courts. It is important to choose a laboratory with an impeccable reputation and a long, error-free service record. A simple, first step to determining how responsive to your needs the laboratory will be is to find out the hours their doctoral-level staff is available to speak with you. Be direct; ask, “If I have a question regarding DNA testing, during what hours are doctoral level staff members from your company available to answer my concerns?” It is also a good idea to ask if there is a charge for such consultations.
Turnaround Time
DNA paternity test results are usually obtainable within anywhere from two days to a week, though 24-hour turnaround may also be available. It is important to remember that DNA family relationship testing involves sophisticated genetic analyses.
Occasionally, genetic mutations are encountered in DNA paternity cases, and certain cases of complicated DNA family reconstructions require expert consultations and additional, specialized testing. An efficient, experienced DNA testing laboratory offers quick two-day turnaround results on most DNA tests they perform, and should be willing to devote their expertise, time and additional resources to more complex cases to achieve the most conclusive results.
Summary
Technological, social, and legal developments have fueled more widespread use of DNA family relation- ship testing services over the past decade. Yet, it remains difficult for both the general public and medical professionals to identify reliable, accredited DNA testing laboratories. As such, partnerships between healthcare providers and reliable DNA testing laboratories may allow patients seeking DNA family relationship testing to simultaneously receive reliable, conclusive DNA test results and competent, professional, clinical care. Due to the entrepreneurial nature of and breadth of services offered in this discipline, urgent care providers may be well positioned to engage in such partnerships.
References
- American Association of Blood Banks, Annual Report Summary for American Association of Blood Banks corporate website, available at: http://www.aabb.org/Content/Accreditation/Parentage_Testing_Accreditation_Program/ptprog.htm. Accessed April 4, 2007.
- Social Services Amendments of 1974, L. No. 93-647, 88 Stat.2337 http://www.aabb.org/Content/Accreditation/Parentage_Testing_Accreditation_Pro- (1975). 3. Family Support Act of 1988, Pub. L. No. 100-485, 102 Stat. 2343 (1988).
- The National Conference of State Available at: http://www.ncsl.org/ programs/cyf/paternitylegis.htm. Accessed April 4, 2007.
- Fathers’ Rights Wikipedia. Available at http://en/wikipedia.org/ wiki/Fathers’_rights. Accessed April 4, 2007.
- Clinical Laboratory Improvement Amendments of 1988, L. No. 100- 578, 102 Stat. 2903 (1988) Title 42 Code of Federal Regulations (CFR), Part 493.3.
- New York State Department of Health Clinical Laboratory Statute and Regulations, Section 58-1.7 of 10NYCRR, available at: http://www.wadsworth.org/ labcert/clep/faq/faq.htm.
- Federal Trade Commission for the At-home genetic tests: a healthy dose of skepticism may be the best prescription, July 2006. available at: http://www.ftc.gov/bcp/edu/pubs/consumer/health/hea02.htm.
- Anderlik Assessing the quality of DNA-based parentage testing: findings from a survey of laboratories. Jurimetrics J. 2003;43:291-314.

